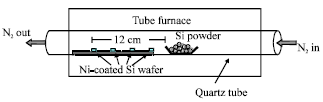
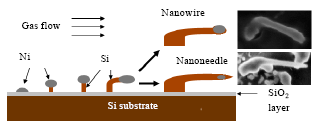
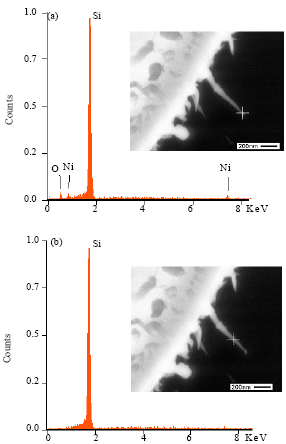
Research Article
One Dimensional Silicon Nanostructures Synthesized via Thermal Evaporation on Nickel Coated Silicon Wafer: Effect of Substrate Position
School of Materials and Mineral Resources Engineering, Universiti Sains Malaysia, 14300 Nibong Tebal, Penang, Malaysia
S. D. Hutagalung
School of Materials and Mineral Resources Engineering, Universiti Sains Malaysia, 14300 Nibong Tebal, Penang, Malaysia