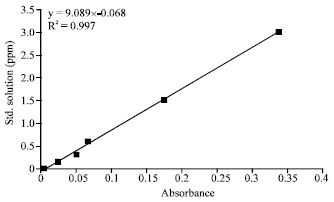
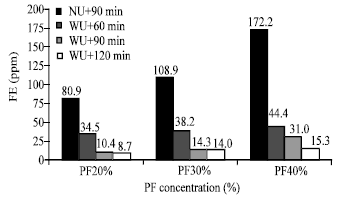
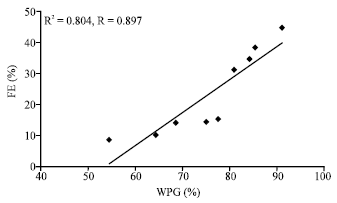
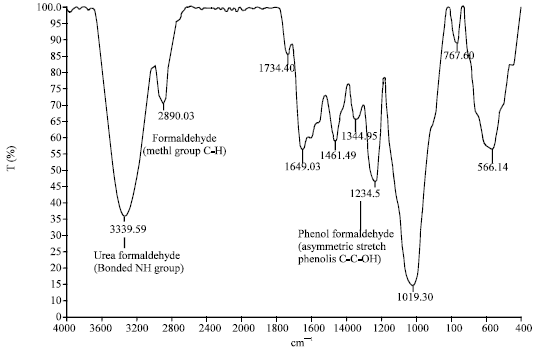
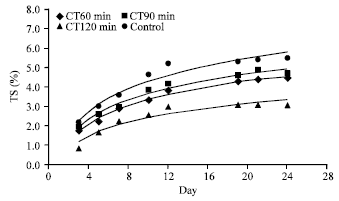
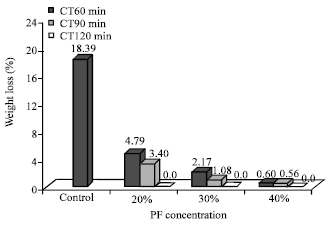
Research Article
Enhancing the Properties of Low Density Hardwood Dyera costulata Through Impregnation with Phenolic Resin Admixed with Formaldehyde Scavenger
Faculty of Forestry, Universiti Putra Malaysia, 43400 Selangor, Malaysia
A. Zaidon
Faculty of Forestry, Universiti Putra Malaysia, 43400 Selangor, Malaysia
M.A. Rabia`tol Adawiah
Faculty of Forestry, Universiti Putra Malaysia, 43400 Selangor, Malaysia
E.S. Bakar
Faculty of Forestry, Universiti Putra Malaysia, 43400 Selangor, Malaysia
M.T. Paridah
Institute of Tropical Forestry and Forest Products, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
S. Mohd. Hamami
Faculty of Forestry, Universiti Putra Malaysia, 43400 Selangor, Malaysia
U.M.K. Anwar
Institute of Tropical Forestry and Forest Products, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia