Research Article
Interaction of Lime and Sodium Chloride in a Low Plasticity Fine Grain Soils
Research Institute for Water Scarcity and Drought, Iran
E. Kabir
Faculty of Civil Engineering, Central Tehran Branch, Iran
Now a day, with increasing civil engineering projects, designers and engineers encounter low resistance lands and are challenged with complex problems more than before. Soil stabilization by lime is one of the common methods of soil improvements, which has been used since long time. In many parts of Iran, especially in south tropical areas, soils contain a significant amount of sodium chloride, which creates a weak ionic interaction between soil grains. However, the existing natural salt may affect the interaction of lime and soil.
In the process of soil strengthening by adding lime, the following four reactions may occur in a soil-lime mixture (Azarfar and Abdi, 2003). The first is exothermic hydration reaction, in which lime reacts with water and quicklime Ca(OH)2 is produced. This reaction happens in short period of time and causes a reduction in the mixture moisture. Released calcium ions by quicklime make starting some ionic interchanges in fine grain soil momentary (Jagannath et al., 2004).
The second reaction of lime and soil is carbonation reaction in which lime interacts with air carbonic gas and cause to get back lime to bicarbonate or get into slaked lime (Kezdi, 1979).
The third reaction is ionic exchange which takes places rapidly in fine grain soils in the presence of water. After increasing the concentration of calcium ions around clay particles due to hydration of lime, the existing low electro-negative ions are replaced by calcium ions and consequently the thickness of double layer reduces which results in a reduction in the space between clay minerals. So, the gravity between minerals is increased which causes a kind of flocculation of particles or lumping to each other (Jagannath et al., 2004). This reaction happens immediately after adding water and lime to soil and cause a significant reduction in the soil plasticity and swelling and also a considerable increase in its competency and strength. Furthermore the texture of clayey soils is changed because clay particles tend to couple with each other and form bigger grains (Puppala et al., 2004).
The fourth reaction is pozzolanic reaction which takes places in the presence of water, with silicates and aluminates and creates sticky gels that have a higher bearing capacity rather than natural soil. Some of fine grain soils, specially clay minerals are naturally pozoolanic and have ability to compose with lime and produce sticky gels. Contrary to ionic interchanges reaction, the pozoolanic reaction is done gradually (Azarfar and Abdi, 2003).
Many attentions have been also paid to the effect of salt on soil properties. According to Petrukhin (1993) chlorine salts cause a decrease in the optimum moisture content and an increase in the maximum dry density of soil. He reported that the presence of soluble salts in loess soil causes the maximum dry density increases up to 10% and the optimum water content decreases between 8 to 16%. Recently, Abood et al. (2007) studied the effects of adding three chloride compounds (NaCl, MgCl2, CaCl2) on the properties of a silty clay soil. The soil was tested for its liquid limit, plastic limit, dry unit weight and moisture content and shears strength. The addition of each chloride compound decreased the liquid limit, the plastic limit, the plasticity index and the optimum moisture content of the soil; while the dry density increased with an increase in salts percentage. The compressive strength of the soil also increased with the addition of chloride compounds and they concluded that these salts could help improving soil strength as well as its physical properties.
Many experiments have been carried out to investigate the possibilities of using various binders including gypsum (Kuno et al., 1989), carbonated-aluminate-salt (Kamon et al., 1989), pulverized fuel ash and blast furnace slag (Hebib and Farrell, 2003) to stabilize organic soils. Not only the types of binders, but also the salt concentration plays an important role in stabilization of organic soil (Onitsuka et al., 2003). It was reported that the adverse effect of organic matter on strength of stabilized soil can be mitigated in soil having high salt concentration (Onitsuka et al., 2003). To extend this finding in an application, binders of lime with addition of sodium chloride (NaCl) and calcium chloride (CaCl2) were examined for organic soil stabilization. Lime was selected as main binder in this chapter because lime was more sensitive with organic than cement (Onitsuka et al., 2003) and recently cement stabilization was found to cause water and soil pollutions by leaching of hexavalent chromium (Tsuneoka et al., 2003).
In terms of effect of soluble salts on soil improvement by lime some researches have been carried out. According to Tremblay et al. (2002) NaCl as one of organic material has a significant effect on the strength gain of lime stabilization due to the obstruction of pozzolanic reaction. However, not all of the organic compounds have a negative effect. But, Davidson et al. (1965) reported sodium chloride salt makes proportional increasing in compressive strength of stabilized samples with lime. He explained this is because of PH increasing due to the presence of NaOH which is produced by the reaction between sodium chloride and lime.
| (1) |
The high basic ambiance causes to increase silicate dissolution for reacting with calcium and pozzolanic process and to produce cement. Furthermore a mixture of calcium-sodium silicate gel improves the quality of cement particles faster than calcium silicate gel.
This research was carried out to examine to what extent the sodium chloride will affect the soil strengthening by adding lime in short and long term.
This research was started in October 2008 in the center of Golestan province, Gorgan city, which a large portion of its territory is covered by saline soils. The study started with providing appropriate soil samples and after a period of eleven months of experimental works was finished in November 2009. CL-ML soil with liquid and plastic limits respectively 25.75 and 20.24 and a plastic index 5.51 and specific gravity 2.65 was used. The maximum dry unit weight and its optimum moisture was 1.76 g cm-3 and 18.28%. Sodium chloride was transported from Sirjan mine and lime was a production of Semnan lime factory with 0.56 g cm-3 specific weight. For surveying the effect of lime on unconfined strength of soil which is containing sodium chloride, the following mixtures were prepared and tested:
| • | Soil with 2, 4, 6 and 8 weight percentage lime |
| • | Soil with 1, 2, 3 and 5 weight percentage sodium chloride |
| • | Soil with 2, 4, 6 and 8 weight percentage lime and 1, 2, 3 and 5 weight percentage sodium chloride conjugate |
For making samples, first all of soils were crushed with plastic hammer after drying in weather. After that, they were passed through sieve No. 10. The composing of lime and sodium chloride with soil are happened lonely or together, in this regular. First they are mixed when they are dry. After that some water based on optimum moisture (18.27) and percentage of dry weight of all material is increased.
For sample making based on proctor mould, the samples were compacted in 3 layers with 2.5 kg standard hammer. After pound ling the soil in simple proctor mould with standard compaction hammer, the sidewall sampler was used in mould for sampling from condensed soil mixture. The sampler pipe was placed on top of mould for drawing out the samples. The soil around the pipes was removed carefully and finally the samples were drawing out with hydraulic jakes. The curing time was 7 and 28 days in laboratory’s temperature and to prevent from getting dry and lose the moisture, they were kept in plastic bag. All tests have repeated in 3 times.
In this research, the following tests were carried out: hydrometery based on standard ASTM D 422-63, specific gravity based on ASTM D 854-87, Atterberg limitat based on ASTM D 4318-87, standard proctor based on ASTM D 698 and uniaxial compressive strength based on ASTM 2166-87.
The specification of all samples and the results of all tests are presented in Table 1. As shown twenty four samples were prepared and tested as described in earlier.
The influence of lime on the compressive strength: Variation of compressive strength of mixture of soil and lime with respect to lime percentage and curing time is shown in Fig. 1. As observed, by adding 2% of lime to the soil the compressive strength of mixture has increased from 67.2 to 95.6 KPa after 7 days and to 236.8 KPa after 28 days. The improvement in resistance after 7 days is due to the cation exchange reaction phenomenon, while the jump in the resistance after 28 days is related to developing the pozzolanic reaction to a high extent. The increase in resistance of samples with a percentage of lime of 4, 6 and 8 is much more than previous sample such that they rise to 154.1, 169.2 and 173.5 KPa after 7 days and to 260.8, 269.2 and 314.8 KPa after 28 days, respectively.
Figure 2 presents the improvement of uniaxial compressive strength in 7th day and 28th day for different lime percentage.
The issue which is important, as revealed in Fig. 1, is that the rate of resistance improvement decreases with an increase in lime percent such that it becomes gentle after 6% of lime; therefore one may propose 6% as the optimum percentage of lime for stabilizing this soil.
The influence of sodium chloride on the compressive strength: The results of uniaxial compressive strength after 7 days curing of samples containing 1, 2, 3 and 5% of sodium chloride are shown in Fig. 3. By increasing 1% sodium chloride to samples after 7 days curing, the resistance of samples is reduced from 67.2 to 25.1 KPa. The compressive strength decreases as the percentage of the salt increases such that it becomes 19, 18.5 and 15.7 KPa after increasing the rate of sodium chloride to 3, 4 and 5%, respectively.
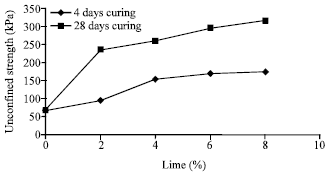 | |
| Fig. 1: | Variation of uniaxial compressive strength with respect to added lime after 7 and 28 days of curing |
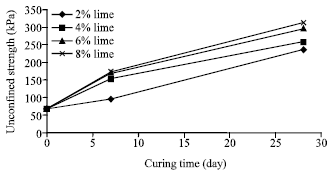 | |
| Fig. 2: | Variation of uniaxial compressive strength versus curing time for different percentage of lime |
| Table 1: | Results of uniaxial compressive strength tests conducted on samples |
 | |
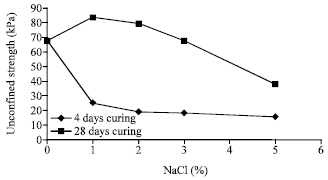 | |
| Fig. 3: | Variation of unconfined compressive strength versus sodium chloride for 7 day and 28 day samples |
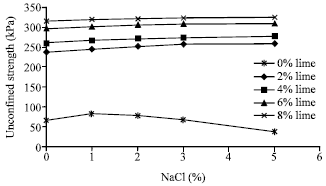 | |
| Fig. 4: | Variation of uniaxial compressive strength of 28 day samples containing lime and sodium chloride |
The decrease in resistance is due to the presence of large amount of Na+ ions around the soil particles which in turn causes the double layer around the clay particles increases. This issue results in an increase in the distance between soil particles and consequently a decrease in the compressive strength of the soil.
The results of compressive strength of samples containing 1, 2, 3 and 5% of sodium chloride after 28 days of curing are shown in Fig. 3. In samples with 1% sodium chloride, the unconfined compressive resistance increases from 67.2 KPa for pure soil to 84 KPa after 28 days of curing. But it decreases as the percentage of the salt increases such that it becomes 79.4, 67.6 and 37.6 KPa after increasing the rate of sodium chloride to 3, 4 and 5%, respectively.
The reason of the increase of strength under 1% of salt is the ionic bands created between soil particles. But in samples containing more salt, the soil becomes rich in Na+ ions which results in soil dispersing and consequently a reduction in the strength. Therefore, in samples having 3% of sodium chloride the unconfined strength is decreased nearly to the pure soil and in samples with 5% of salt, the large amount of sodium ions are so much that its dispersing effect is dominant to its ionic connection effect, therefore the unconfined strength drops down to somewhat much less than the pure soil and close to 7 day samples.
 | |
| Fig. 5: | Variation of uniaxial compressive strength of 7 day samples containing lime and sodium chloride |
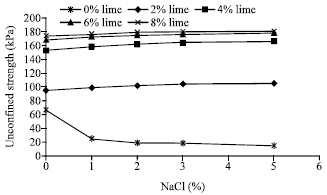
The effect of sodium chloride on unconfined strength of stabilized samples with lime: The results of compressive strength test of samples containing 2, 4, 6 and 8% lime conjugate with 1, 2, 3 and 5% sodium chloride after 28 days of curing are shown in Fig. 4. As revealed, regardless the percentage of lime, the presence of sodium chloride has a positive effect on the unconfined compressive strength of the soil. Moreover, its influence increases as its percentage increases, but this effect will be negligible after almost 5% of concentration.
In samples with 2% lime by adding 1 to 5% of sodium chloride, the strength increases about 4 to 9%. With increasing the percentage of lime the influence of salt is reduced such that in samples containing 8% lime by adding 1 to 5% sodium chloride, the strength improvement is limited between 1 to 3%. One may conclude that in samples containing high percentage of lime, the influence of pozoolanic reaction is much enough to dominant the influence of salt. However, in samples containing 6 and 8% of lime, the presence of sodium chloride has resulted in 1 to 4% increase in strength.
The effect of sodium chloride on unconfined strength after 7 days of curing is presented in Fig. 5. In these samples, the effect of sodium chloride is similar to 28 day samples but slightly more. For example in samples containing 2% of lime, by adding up to 5% sodium chloride, the resistance of samples increases up to 10%.
As shown, the efficiency of lime on stabilization of saline soils is slightly more than non-saline soils. However, one may conclude that in spite sodium chloride reduces the compressive strength of pure soil significantly, but the presence of lime in the soil results in inversing this effect and causes a slight additional improvement in soil strength.
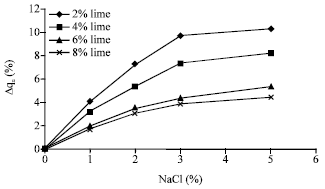 | |
| Fig. 6: | Relative increased compressive strength of samples containing lime versus sodium chloride (after 7 days curing) |
For 28 day samples, as presented in Fig. 4, similarly to 7 day samples, the performance of lime on stabilization of salty soils is slightly better than non-saline soils. Also it is concluded that in spite the effect of sodium chloride on the compressive strength of normal soils is a function of its concentration, but the presence of lime results in a slight additional improvement in soil strength with a continuous trend.
For a better understanding, relative increase in unconfined compressive strength of samples are shown in Fig. 6 and 7 for 7 and 28 day samples, respectively. As definition relative increase in unconfined compressive strength is obtained from Eq. 2 in which q0 is unconfined compressive strength of soil without sodium chloride and Δqu is the increased unconfined compressive strength due to the presence of sodium chloride.
| (2) |
As revealed, both of 7 and 28 day samples present similar behaviors with respect to sodium chloride, i.e. the higher the percentage of salt, the more is the relative increase in compressive strength. However, the rate of rising in relative increased strength is a function of salt concentrations such that this rate is less pronounced for concentrations more than 3%. In the other hand, by increasing the amount of lime, the effect of sodium chloride is less pronounced.
These results are well matched with the results reported by Azarfar and Abdi (2003), however, the slight difference of increased strength observed between their results and those presented in this research is attributed to the differences in the characteristics of soils used by them.
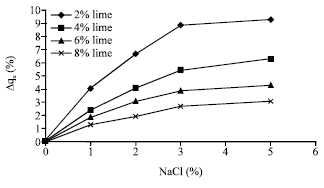 | |
| Fig. 7: | The relative increased compressive strength of samples containing lime versus sodium chloride (after 28 days curing) |
These results are also in a good agreement with the theory of Davidson et al. (1965). So that the increased resistance in stabilized soil with lime in the presence of sodium chloride, is attributed to an increase in PH due to the presence of NaOH which is explained by the reaction 1.
As it's well known, the pozzolanic reaction which is mainly consisted of interaction between lime, water, silicates and aluminates is a function of the solubility of silicates and aluminates which in turn is a function of PH of the media. That's why the presence of sodium chloride improves the performance of lime in soil strengthening (Kabir, 2009).
In a comparison, the results of this study confirm the findings of Abood et al. (2007) reporting an increase in compressive strength of soil due to the presence of soluble chloride salts. Also the report of Onitsuka et al. (2003) regarding the improvement of organic soil by high concentration of salt is approved. However, the findings of this research is completely in contradiction with Tremblay et al. (2002) which believe NaCl spoils the effect of lime in soil strengthening.
Based on the results of tests carried out on the samples prepared by mixing CL-ML soil with lime and sodium chloride, the following points are concluded:
The unconfined strength of soil increases by increasing the percentage of lime
The unconfined strength of soil strengthened by lime improves by increasing the curing time. Poozoolanic reaction which is a time dependent one is the governing parameter of this phenomenon.
Comparing the rate of increase in unconfined strength of samples mixed with different percentage of lime after 28 curing days, it's revealed that the optimum magnitude of lime is 6%.
Sodium chloride curing causes a large reduction in 7 day resistance such that 1% sodium chloride causes 63% reduction in the soil resistance. Similarly, in samples containing 2, 3 and 5% of sodium chloride exhibit 72, 72.5 and 77% decrease in strength, respectively. This influence is attributed to accumulation Na+ ions around the clay’s material and increasing the density of double layer and so scattering of soil particles.
Small amounts of sodium chloride improve the 28 day strength of soil, but it decreases if the quantity of salt exceeds 3%. The results show an increase of 25 and 18% in unconfined resistance in samples containing 1 and 2% of salt, while at 3% sodium chloride, the resistance is equal to natural soil and for soil containing 5% sodium chloride the compressive strength drops 44%.
One may attribute this behavior to ionic bonds between sodium chloride and soil minerals. Low values of sodium chloride provides a small amount of Na+ between soil grains which create weak ionic connections between them, while with large quantities of salt Na+ surround soil minerals and create a thick double layer which result in dispersing soil particles and thus reduce the shear strength and unconfined strength.
Adding sodium chloride to a mixture of soil and lime improves the compressive strength of mixture. This increase is due to an increase in pH of the media by sodium chloride and thus an increase in the dissolubility of silicates to interact with calcium and create poozoolanic process to produce cement. Also a calcium-sodium silicate gel improves the cementation faster than a calcium silicate gel.
In samples containing large amount of lime, i.e., 6-8%, the effect of sodium chloride is less than those containing small quantity of lime, because the soil resistance has been already promoted by lime.
Based on this research, it is observed that existence of certain percentage of sodium chloride in CL-ML soils causes the compressive resistance increases somewhat about 10% and thus inversely, water leaching and desalinization may weaken saline soils to a degree of about 10%.