Research Article
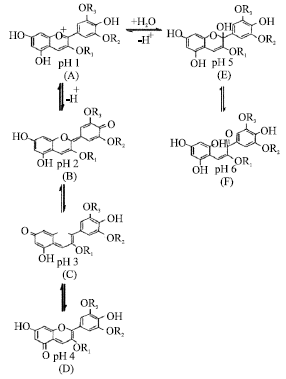
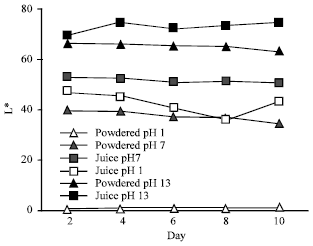
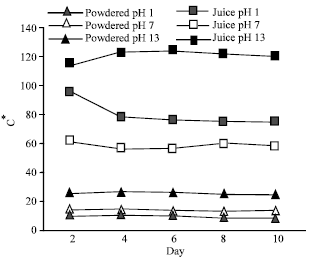
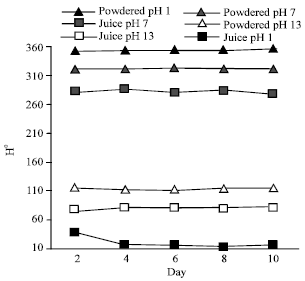
The Effect of pH on Color Behavior of Brassica oleracea Anthocyanin
Faculty of Chemical Engineering, Universiti Teknologi MARA (UiTM), 40450 Shah Alam, Selangor, Malaysia
Ida Idayu Muhammad
Department of Bioprocess Engineering, Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia (UTM), 81310 Skudai, Johor, Malaysia
Ruzitah Mohd Salleh
Faculty of Chemical Engineering, Universiti Teknologi MARA (UiTM), 40450 Shah Alam, Selangor, Malaysia