Research Article
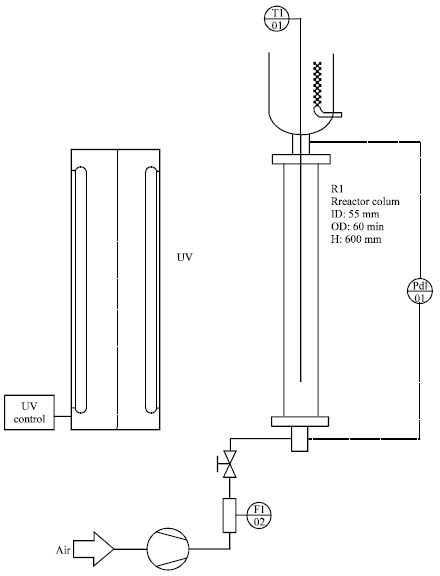
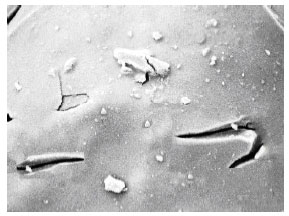
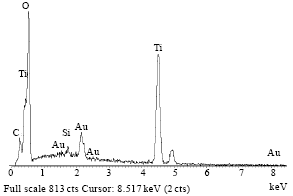
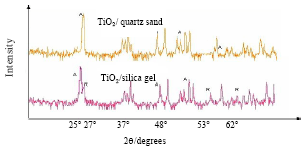
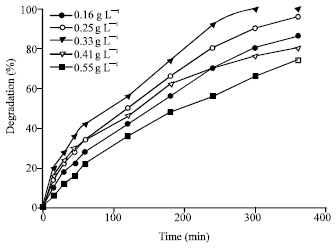
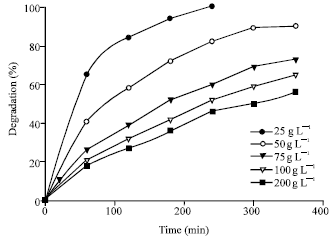
Photocatalytic Degradation of Phenol in a Fluidized Bed Reactor Utilizing Immobilized TiO2 Photocatalyst: Characterization and Process Studies
School of Bioprocess Engineering, Universiti Malaysia Perlis, Kompleks Pusat Pengajian Jejawi 3 02600 Jejawi, Perlis, Malaysia
N. A. H. Hairul
School of Bioprocess Engineering, Universiti Malaysia Perlis, Kompleks Pusat Pengajian Jejawi 3 02600 Jejawi, Perlis, Malaysia
H. M. R. Akmal
School of Bioprocess Engineering, Universiti Malaysia Perlis, Kompleks Pusat Pengajian Jejawi 3 02600 Jejawi, Perlis, Malaysia
M. Abdul Rahman
School of Chemical Engineering, Engineering Campus, Universiti Sains Malaysia, 14300 Nibong Tebal, Penang, Malaysia