Research Article
Effect of Vermiculite Addition on Thermal Characteristic of Water-based Acrylic Fire Retardant Coating Formulation
Department of Mechanical Engineering, Universiti of Teknologi, PETRONAS, Bandar Seri Iskandar, 31750, Tronoh, Perak, Malaysia
Bambang Ariwahjoedi
Department of Fundamental Applied Science, Universiti of Teknologi, PETRONAS, Bandar Seri Iskandar, 31750, Tronoh, Perak, Malaysia
Patthi Hussain
Department of Mechanical Engineering, Universiti of Teknologi, PETRONAS, Bandar Seri Iskandar, 31750, Tronoh, Perak, Malaysia









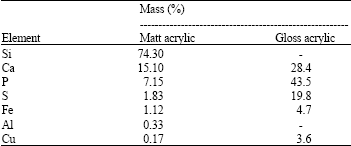

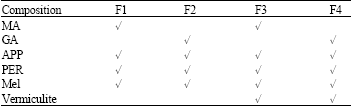
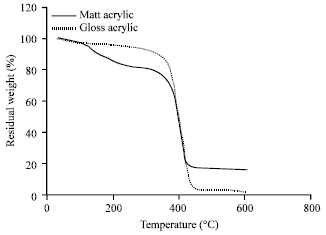
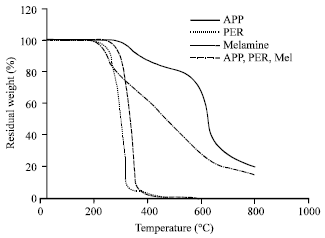
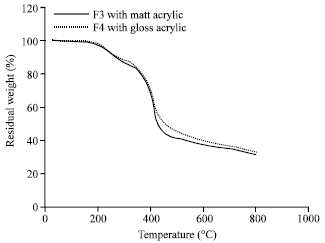


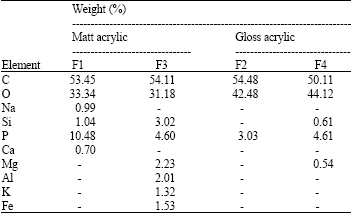
ZAHIR PASHA Reply
who are the manufacturers of this fire retardant paint. Are there any producers of this paint in the UAE.