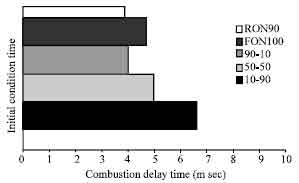
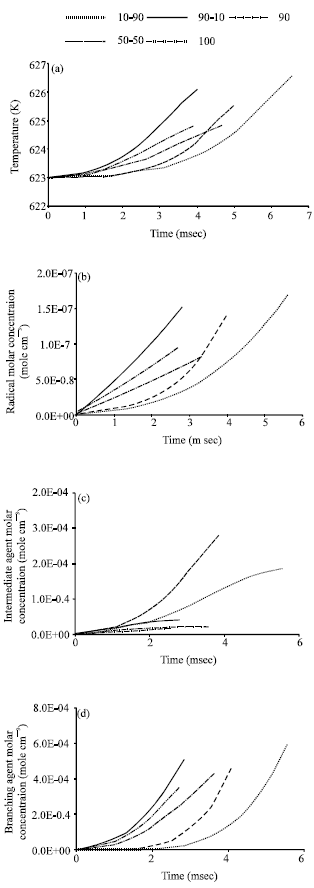
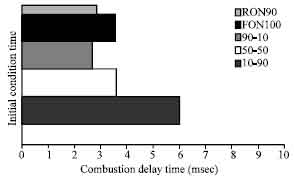
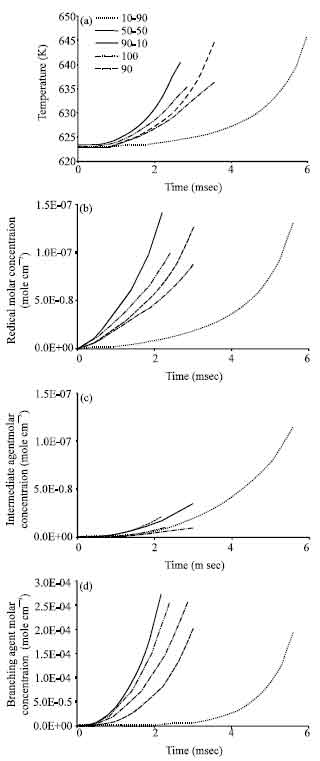
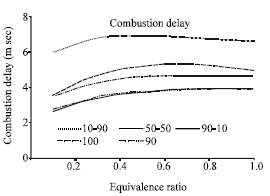
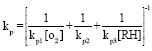Research Article
The Combustion Behavior Analysis of Dual Fuel HCCI using the Shell Model
Center for Automotive Research, Department of Mechanical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
A. Aziz
Center for Automotive Research, Department of Mechanical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia