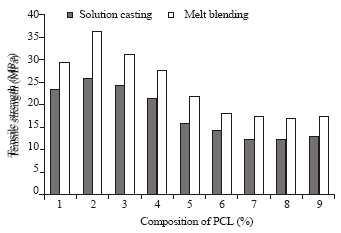
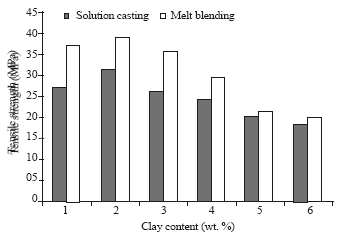
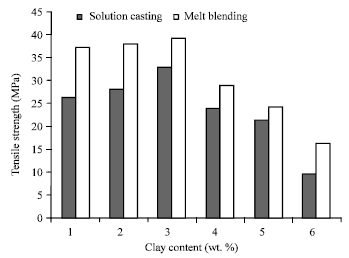
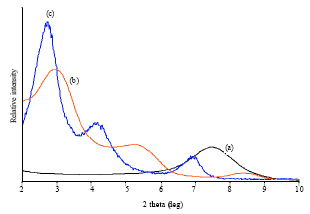
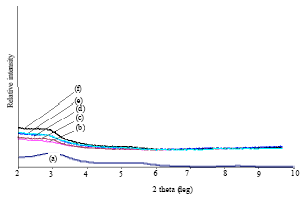
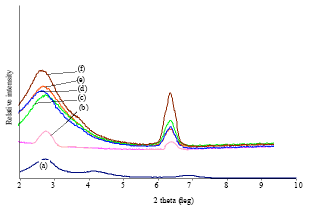
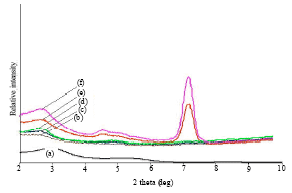
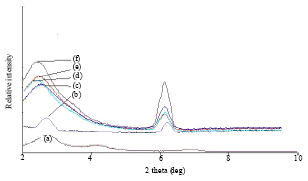
Research Article
Preparation and Characterization of Polylactic Acid/Polycaprolactone Clay Nanocomposites
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia 43400, Serdang, Selangor, Malaysia
Mansor B. Ahmad
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia 43400, Serdang, Selangor, Malaysia
Emad A. Jaffar Al-Mulla
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia 43400, Serdang, Selangor, Malaysia
Nor Azowa Bt Ibrahim
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia 43400, Serdang, Selangor, Malaysia