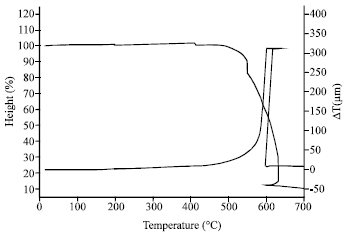
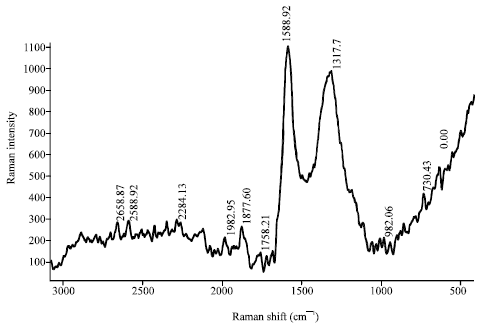
Research Article
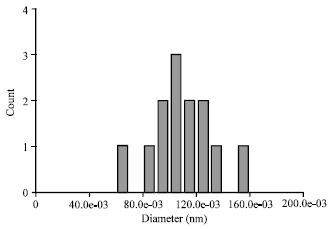
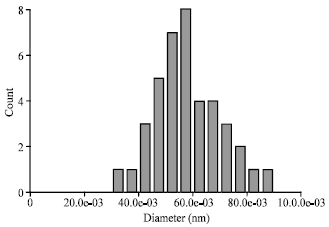
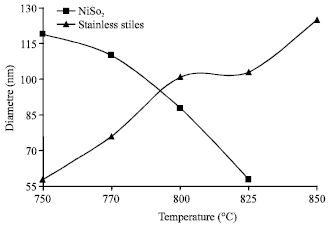
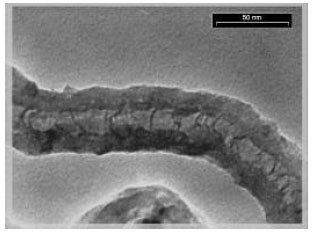
Comparison of Synthesis and Purification of Carbon Nanotubes by Thermal Chemical Vapor Deposition on the Nickel-Based Catalysts: NiSio2 and 304-Type Stainless Steel
Department of Physics, Sharif University of Technology, P.O. Box 11365-9161, Tehran, Iran
A. Jafari
Department of Physics, Guilan University of Technology, P.O. Box 4193833-697, Rasht, Iran
S. Fekri
Tabriz University, P.O. Box 5166616-471, Tabriz, Iran
A. Shafikhani
Physics Group, Azahra University, P.O. Box 19938, Tehran, Iran
M. Vesaghi
Department of Physics, Sharif University of Technology, P.O. Box 11365-9161, Tehran, Iran
G. Behzadi
Islamic Azad University, East Tehran Branch (Ghyamdasht), P.O. Box 33955-163, Tehran, Iran