ABSTRACT
The goal of this study was to determine whether orexin affects the mean plasma concentrations of metabolic parameters such as thyroxine (T4), triiodothyronine (T3), Growth Hormone (GH), insulin, glucagon, glucose, fatty acid and urea in the goats fed different energy content in diets. Sixteen goats were randomly divided into 4 groups. Animals in group 1 and 2 were fed 100% and animals in groups 3 and 4 were fed 50% energy content in diet for 20 days. After 20 days, animals in groups 1 and 3 received daily infusion of 1 μg orexin and groups 2 and 4 received daily infusion of 2 μg orexin into their third ventricle for 5 days. Lower dietary energy intake and infusions of 1 and 2 μg orexin significantly (p<0.01) decreased the mean plasma concentrations of T3, T4, insulin and glucose and significantly (p<0.01) increased the mean plasma concentrations of GH, glucagon, fatty acid and urea of the animals in groups 3 and 4. The results of this experiment indicated that orexin may negatively affect the T3, T4, insulin and glucose and increase GH, glucagon, fatty acid and urea in the goats with negative energy balance, but not in those with the positive energy balance.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2010.644.651
URL: https://scialert.net/abstract/?doi=jas.2010.644.651
INTRODUCTION
Orexin is a 33-amino-acid neuropeptide that is mostly found in hypothalamus (Antunes et al., 2001; Arihara et al., 2000; Backberg et al., 2002). Based on its neuron distributions in hypothalamus, orexin coexists with many other neurons. For example, in hypothalamic area, orexin coexists with neurons secreting different neurotransmitters such as GHRH, GABA, noradrenaline, 5-hydroxytryptamine (5-HT) and NPY (Al-Barazanji et al., 2001; Balasko et al., 1999). Therefore, orexin controls different physiological actions on many different tracts (Antunes et al., 2001; Al-Barazanji et al., 2001; Arihara et al., 2000; Backberg et al., 2002; Balasko et al., 1999; Ehmke and Just, 2003). One of the physiological actions is its effect on metabolism and feeding behaviors that make orexin as an orexinergic hormone (Backberg et al., 2002). The orexigenic effect of orexin decrease or increase plasma levels of insulin, glucagon, somatostatin, gastrin and increase the release of growth hormone (Hagan et al., 1999; Lopez et al., 2004; Meghan and Samson, 2003; Mitsuma et al., 1999; Nowak et al., 2005; Russell et al., 2002). Most of the above studies were conducted in human and rat as a nonruminant. Ruminants have different metabolism from that of nonruminants (Harrison and Leat, 1975). It is assumed that the control of feeding behavior is different from that of nonruminants. There are very few reports about the orexigenic effect of orexin on the metabolic hormones in ruminants fed different energy content in diet. Therefore, the first goal of this experiment was to determine whether orexin affects the mean concentrations of metabolic parameters in the goats fed different energy content in diets.
Among many studies done on the effect of orexin on metabolic hormone, there are no reports about the effect of the orexin on thyroid hormones under different energy intake. The importance of thyroid hormones in metabolism is well known. For example, thyroid hormones play an important role in the regulation of energy homeostasis via oxygen consumption and heat generation (De Jesus et al., 2001; Lanni et al., 2001). Changes in basal metabolic rate caused by different energy content in diet is accompanied by changes of thyroid hormones secretions. Therefore, the second goal of this study was to determine whether orexin alters the thyroid hormones secretion in the goats fed different energy content in diet.
MATERIALS AND METHODS
Experimental design: This experiment were conducted during 2007-2009. Sixteen goats (weighing between 40 to 50 kg) were randomly divided into 4 groups and kept in Animal Science Research Institute of Karaj. Animals in group 1 and 2 were fed 100% energy (NE) and animals in group 3 and 4 were fed 50% energy (LE) content in diet for 20 days. Gross energy and chemical compositions of feedstuffs consisted of dry mater, crude protein, crude fiber, ether extract, total ash, NDF, ADF, calcium and phosphorous were analyzed in the Animal Science Research Institute of Karaj during 2007-2009.
| Table 1: | Experimental rations and prepared energy and nutrients |
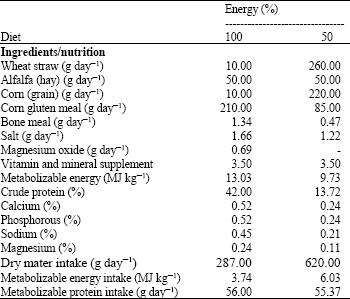 | |
Diets were formulated based on (Agricultural and Food Research Council, 1995) (Table 1). We chose this formulation based on the Shahid Beheshti University permission. During the course of the experiment, daily feed was weighed based on body weight and individually given to each goat every morning. The goats had free access to fresh water. Diet 1 and 2 were consisted 100 and 50% of maintenance energy requirements, respectively. Other requirements were balanced at maintenance level. After 20 days, all animals were prepared for surgery. Goats were anesthetized throughout the surgery for third ventricle cannulation under stereotaxic methods and jugular vein cannulations. Surgical procedures were done under general anesthesia induced by sodium pentobarbital and maintained by halothane in a closed circuit system (Khazali, 1992). Each goat was kept in a single cage for a 4 days recovery period. During recovery period, cannules were washed by PBS solution to prevent from clotting. After surgery, on day 5, goats in group 1 and 3 received 1 μg orexin and goats in group 2 and 4 received 2 μg orexin into their third ventricles for 5 days. Body weight of animals was measured on day 1 and 20 of the experiment.
Blood collection: Blood samples were collected from cannules that were put into the jugular veins, everyday from 4 days before first infusion of orexin until 4 days after the last orexin infusion. Blood samples were kept at 4°C until centrifugation. A saturated sodium citrate solution (40 μL sodium citrate solution mL-1 blood) was added to the samples before centrifugation to prevent clotting of plasma during storage. Plasma was stored at -20°C until assayed for T3, T4, insulin, GH, glucagon, glucose, fatty acid and urea.
Hormone assays: Plasma T3, T4, insulin, GH and glucagon were measured by a homologous double-antibody radioimmunoassay (RIA). For GH assay, ovine GH (TYN-OG) and antisera against GH were provided by Tabeshyarnoor Co.(Industerial City of Bu-Ali, Hamadan, Iran). Ovine GH (TYN-OG) was used for iodination. A seven-point standard curve ranging from 0.04 to 10 ng GH was used. An average assay binding of 40% was achieved using an initial 1:20000 dilution of GH antiserum for GH assays. The inter- and intra-assay variations were 6 and 9%, respectively. For insulin assay, ovine insulin (TYN-OI) and antibody against insulin were provided by Tabeshyarnoor Co. (Induterial City of Bu-Ali, Hamadan, Iran). Ovine insulin (TYN-OI) was used for iodination. A seven-point standard curve ranging from 0.02 to 10 ng insulin was used. An average assay binding of 30% was achieved using an initial 1:5000 dilution of insulin antiserum for insulin assays. The inter- and intra-assay variations were 8 and 5%, respectively. For glucagon assay, human glucagon (TYN-HC) and antibody against glucagon were provided by Tabeshyarnoor Co. (Industerial City of Bu-Ali, Hamadan, Iran). Human glucagon (TYN-HC) was used for iodination. A seven-point standard curve ranging from 0.02 to 10 ng insulin was used. An average assay binding of 35% was achieved using an initial 1:10000 dilution of glucagon antiserum for glucagon assays. The inter- and intra-assay variations were 7 and 6% respectively. For T3 assay, T2 was purchased from Sigma Chemical Company and T3 antisera were purchased from Chemicon Co. (Temmecula, Ca). The T2 was used for iodination. A six-point standard curve ranging from 0.32 to 5.2 ng T3 mL-1 was used. An average assay binding of 70% was achieved using an initial 1:5000 dilution of T3 antiserum for T3 assays. The inter- and intra-assay variations were 7 and 7%, respectively. For T4 assay, T3 was purchased from sigma chemical company and T4 antisera was purchased from Chemicon Co. (Temmecula, Ca). The T3 was used for iodination. A six-point standard curve ranging from 2.2 to 25 ng T4 mL-1 was used. An average assay binding of 60% was achieved using an initial 1:5000 dilution of T4 antiserum for T4 assays. The inter- and intra-assay variations were 7 and 5%, respectively. For glucose assay, ELISA kits were purchased from sigma chemical company. A six-point standard curve ranging from 20 to 250 mg glucose dL-1 was used. An average assay binding of 35% was achieved. The inter- and intra-assay variations were 4 and 6%, respectivity. For fatty acid assay, ELISA kits were purchased from Sigma Chemical Company. A six-point standard curve ranging from 10 to 150 mg fatty acid dL-1 was used. An average assay binding of 45% was achieved. The inter- and intra-assay variation were 5 and 8%, respectively. For urea assay, ELISA kits were purchased from sigma chemical company. A six-point standard curve ranging from 10 to 150 mg urea dL-1 was used. An average assay binding of 32% was achieved. The inter- and intra-assay variations were 4 and 6%, respectively.
Statistical analysis: All analysis were conducted using General Linear Model procedures (SAS, 1996). Data were analyzed using an analysis of variance for a repeated measure design. Mean comparisons were evaluated by least significant difference with single degree of freedom.
RESULTS
T3 and T4: Infusions of 1 and 2 μg orexin into third ventricle significantly (p<0.01) decreased mean plasma concentrations of T3 , but not T4 of the animals in groups 1 and 2 that were fed NE. Mean plasma T3 levels of the animals in group 1 and 2 were about 2.5, 1.8, 2.2 and 2.4, 1.7, 2.1 ng mL-1 before, during and after infusion of orexin, respectively (Fig. 1). Also, Mean plasma concentrations of T4 of the NE animals in group 1 and 2 were about 41, 40, 40 and 39, 42, 41 ng mL-1 before, during and after infusion of orexin respectively (Fig. 2). Plasma T3 and T4 levels of LE fed animals in groups 3 and 4 were significantly (p<0.01) lower than that of the NE fed animals (Fig. 1, 2).
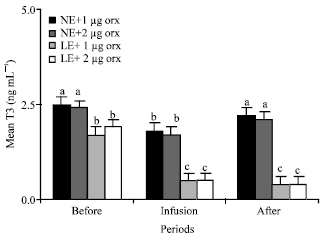 | |
| Fig. 1: | Mean plasma concentrations of T3 of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 μg orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
Orexin infusions significantly (p<0.01) decreased plasma T3 and T4 levels in the LE fed animals (Fig. 1, 2).
GH: Low energy content in diet increased the GH plasma levels of the animals in group 3 and 4 in comparison with plasma GH levels of those animals fed NE. Further to the effect of lower energy dietary intake, infusions of 2 μg orexin significantly (p<0.01) increased the mean plasma GH levels in the animals of group 4 (from 3 to 5), followed by declining GH level from 5 to 4.2 after infusion of orexin. Infusions of 1 μg orexin did not change the mean plasma concentrations of the GH in the animals of group 1 and 2 that fed 100% energy content in diets for 20 days. Mean plasma concentrations of the GH of group 1 were about 1.9, 1.8 and 1.8 ng mL-1 before, during and after infusion of orexin, respectively (Fig. 3). Two micrograms orexin did not change the mean plasma concentrations of the GH in the animals of group 2 that were fed NE. Mean plasma concentrations of the GH of group 2 were about 1.8, 1.7 and 1.7 ng mL-1 before, during and after infusion of orexin, respectively (Fig. 3).
Insulin: Infusions of 1 and 2 μg orexin did not change the mean plasma concentrations of the insulin in the animals of groups 1 and 2 that were fed NE. Mean plasma concentrations of the insulin of the animals in groups 1 and 2 were about 39, 40, 44 and 38, 38, 44 ng mL-1 before, during and after infusion of orexin, respectively (Fig. 4).
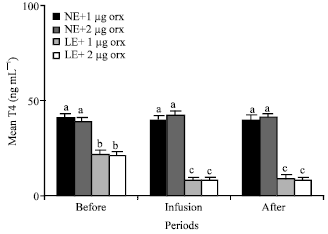 | |
| Fig. 2: | Mean plasma concentrations of T4 of the animals in the different groups of 1 (NE and 1 ug orexin), 2 (NE and 2 ug orexin), 3 (LE and 1 ug orexin) and 4 (LE and 1 ug orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
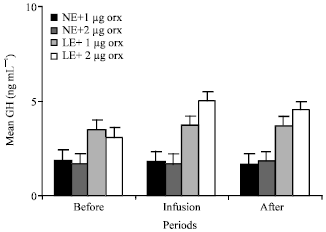 | |
| Fig. 3: | Mean plasma concentrations of GH of the animals in the different groups of 1 (NE and 1 ug orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 ug orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy |
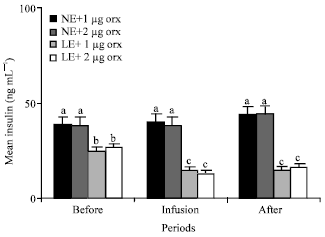 | |
| Fig. 4: | Mean plasma concentrations of insulin of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 ug orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
Mean plasma concentrations of insulin of the animals in groups 3 (25 ng mL-1) and 4 (27 ng mL-1) fed LE were significantly (p<0.01) lower than the plasma insulin levels of those animals in groups 1 (39 ng mL-1) and 2 (38 ng mL-1) fed NE (Fig. 4). Infusions of 1 μg orexin significantly (p<0.01) decreased the mean of the plasma levels of insulin in the animals of group 3 from 25 to 15. Also, mean of the plasma levels of insulin of the animals in group 4 significantly (p<0.01) decreased from 27 to 13 by infusion of 2 μg orexin (Fig. 4).
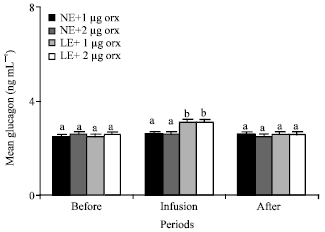 | |
| Fig. 5: | Mean plasma concentrations of glucagon of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 ug orexin), 3 (LE and 1 μg orexin) and 4 (LE and 1 ug orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
Glucagon: Orexin infusions did not change the mean plasma concentrations of the glucagon in the animals of group 1 and 2 that were fed NE. Mean plasma levels of the glucagon of the animals in group 1 and 2 were about 2.5, 2.6, 2.6 and 2.6, 2.6. 2.5 ng mL-1 before, during and after infusion of orexin, respectively (Fig. 5). Infusions of 1 μg orexin significantly (p<0.01) increased the mean of plasma levels of orexin in the animals of group 3 from 2.6 to 3.1, followed by decreasing of glucagon plasma levels from 3.1to 2.6 after infusion of orexin. Also, mean of plasma levels of glucagon of the animals in group 4 significantly (p<0.01) increased from 2.5 to 13.1 by infusion of 2 μg orexin (Fig. 5).
Glucose: Orexin did not change the mean plasma concentrations of the glucose of the animals in group 1 and 2 that were fed NE. Plasma concentrations of the glucose of groups 1 and 2 were about 50, 48, 50 and 52, 50, 50 mg dL-1 before, during and after infusion of orexin, respectively (Fig. 6). Plasma glucose levels of the LE fed animals in groups 3 (42 mg dL-1) and 4 (41 mg dL-1) were significantly (p<0.01) lower than the mean plasma concentrations of glucose of those animals in the group 1 (50 mg dL-1) and 2 (52 mg dL-1) fed NE (Fig. 4). Infusions of 1 and 2 μg orexin significantly (p<0.01) decreased the glucose levels among those animals of group 3 and 4 fed LE (Fig. 6).
Fatty acid: Infusions of 1 and 2 μg orexin significantly (p<0.01) increased the mean plasma concentrations of the fatty acid of the animals in groups 1 and 2 that were fed LE.
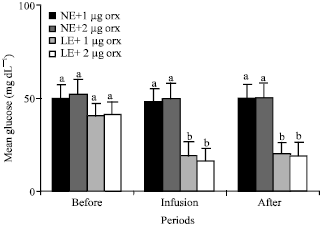 | |
| Fig. 6: | Mean plasma concentrations of glucose of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 μg orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
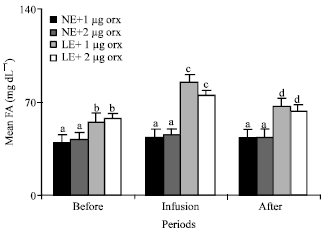 | |
| Fig. 7: | Mean plasma concentrations of fatty acid of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 μg orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
Mean plasma concentrations of fatty acid of the animals in the group 3 (55 mg dL-1) and 4 (57mg dL-1) fed LE were significantly (p<0.01) higher than the mean plasma concentrations of fatty acid of those animals in groups 1 (40 mg dL-1) and 2 (42 mg dL-1) fed NE (Fig. 7). Infusions of 1 and 2 μg orexin significantly (p<0.01) increased the fatty acid levels among those animals fed LE (Fig. 7).
Urea: Orexin significantly (p<0.01) increased the mean plasma concentrations of the urea of the animals in all groups.
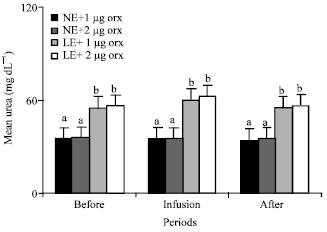 | |
| Fig. 8: | Mean plasma concentrations of urea of the animals in the different groups of 1 (NE and 1 μg orexin), 2 (NE and 2 μg orexin), 3 (LE and 1 μg orexin) and 4 (LE and 1 μg orexin) and before, during and after infusions of ghrelin. NE: Normal energy; LE: Low energy. Treatments with different letter(s) are different at p<0.01 |
Mean plasma concentrations of urea of the animals in group 3 and 4 fed LE were significantly (p<0.01) higher than the mean plasma concentrations of urea of those NE fed animals in groups 1 and 2 (Fig. 8).
Body weight: Low energy dietary intake for 20 days significantly (p<0.01) decreased the mean body weight of the animals from 47 to 35 kg. This was similar to our previous finding reported that negative energy balance decrease body weight in the ewes (Towhidi et al., 2007).
DISCUSSION
T3 and T4: Present study is the first to report the effect of orexin into the third ventricle on thyroid hormones in the ruminants. The results of the effect of orexin on mean plasma T3 and T4 levels of the goats fed LE is different from the previous finding of (Jones et al., 2001; Mitsuma et al., 1999; Russell et al., 2001, 2002) that reported the central effect of orexin decreased the plasma level of Thyroid Stimulating Hormones (TSH) in nonruminants such as rat, but there was no data on the plasma level of T3 and T4 in that study. It is well established that decrease of plasma TSH level is accompanied by increase of plasma T3 and T4 in NE fed nonruminant (Felig and Frohman, 2001; Reasner and Ralbert, 2002). Present results indicate that the NE fed goats as a ruminant response differently to orexin as that of in nonruminant. Only when the ruminant animals are in long term fasting period, are sensitive to the effect of orexin on plasma T3 and T4 levels. The Hypothalamus Pituitary Thyroid (HPT) axis plays important role in the regulation of energy homeostasis (De Jesus et al., 2001; Lanni et al., 2001) via the effects of thyroid hormone to increase oxygen consumption and heat generation (De Jesus et al., 2001; Lanni et al., 2001). Thus, inhibition of the HPT axis during fasting would appear to be an important adaptive mechanism to conserve energy stores (Rondeel et al., 1992; Van Haasteren et al., 1995; Legradi et al., 1997). The state of central hypothyroidism induced by fasting is orchestrated by changes of circulating levels of orexin, which rise with fasting and is restored to normal levels by refeeding (Rondeel et al., 1992). Thus, if orexin iss administered exogenously to fasting animals, the more decrease in circulating levels of thyroid hormones can be observed (Legradi et al., 1998).
GH: Present study is also the first to report the effect of orexin into the third ventricle on GH in the ruminants fed LE. Our result about the effect of orexin on GH in the goats fed LE is different from other studies indicated that orexin decreased GH secretion and enhances the GH response to somatostatin in NE fed nonruminant (Hagan et al., 1999; Lopez et al., 2004; Overeem et al., 2003; Willie et al., 2002). Furthermore, conflicting evidence exist in vitro about the direct effect of orexin on GH, with an no influence on GH secretion observed in rat (Samson and Taylor, 2001). This may be due to decreased plasma level of insulin and glucose (Holl et al., 1999) in the goats fed LE.
Insulin: Our data are different from the studies in nonruminants that indicated orexin has no effect or increase the plasma level of insulin (Meghan and Samson, 2003; Nowak et al., 2005). In those studies, the effect of orexin was not on the long term fasting subject. The mechanism of inhibitory effect of orexin on insulin release most likely maybe like galanin that occurs through the inhibition of adenylate cyclase, involving a petussis-toxin-sensitive inhibitory GTP-binding regulatory protein and the activity of protein kinase C and cyclic AMP (Amiranoff et al., 1988; Lindskog and Ahren, 1991).
Glucagon: Present results are different from the previous studies that reported orexin inhibit glucagon secretions in vitro (Goncz et al., 2008; Quedraogo et al., 2003). This may be due to the plasma glucose concentrations (Manabe et al., 2003). The above study were conducted to determine the effect of orexin on glucagon in vitro. In present study, decreased plasma level of glucose caused by lower energy intake (Marsoobian et al., 1995) and orexin infusions may be the reason for increase level of glucagon and decrease level of insulin.
Glucose, fatty acid and urea: It is well established that low energy content in diet decreases mean plasma concentrations of glucose in most mammals (Marsoobian et al., 1995) as we observed in the goats fed LE. Also, there is a negative correlation between orexin infusion and mean plasma level of glucose in the fasted ruminants whereas in other study is reported that there is a positive correlation between these two parameters in nonfasted nonruminant (Leibowitz et al., 1998).
Present result bout the effect of orexin on urea in the LE fed goats is similar to other study done in the nonruminant that indicated low energy diet increased plasma urea level (Khazali, 1992).
When energy intake is inadequate, proteins can serve as an energy source and plasma urea level is considered as an endproduct of protein catabolism (Ruiz et al., 1971).
Implication: The results of our studies indicated that the third ventricle infusion of orexin may increase the plasma levels of GH, glucagons, fatty acid and urea and decrease the plasma levels of T3, T4, insulin and glucose in the goats with severe body loss. The effect of orexin infusion into third ventricle on metabolic parameters is different from the effect of orexin injections in peripheral circulation. Also, different metabolic system of ruminant and nonruminant animals offer different changes of metabolic status under orexin effect.
REFERENCES
- Al-Barazanji, K.A., S. Wilson, J. Baker, D.S. Jessop and M.S. Harbuz, 2001. Central orexin-A activates hypothalamic-pituitaryadrenal axis and stimulates hypothalamic corticotropin releasing factor and arginine vasopressin neurones in conscious rats. J. Neuroendocrinol., 13: 421-424.
Direct Link - Antunes, V.R.,G.C. Brailoiu, E.H. Kwok, P. Scruggs and N.J. Dun, 2001. Orexins/hypocretins excite rat sympathetic preganglionic neurons in vivo and in vitro. Am J Physiol. Regul. Integr. Comp. Physiol., 281: 1801-1807.
Direct Link - Arihara, Z., K. Takahashi, O. Murakami, K. Totsune and M. Sone et al., 2000. Orexin-A in the human brain and tumor tissues of ganglioneuroblastoma and neuroblastoma. Peptides, 21: 565-570.
Direct Link - Backberg, M., G. Hervieu, S. Wilson and B. Meister, 2002. Orexin receptor-1 (OX-R1) immunoreactivity in chemically identified neurons of the hypothalamus: Focus on orexin targets involved in control of food and water intake. Eur. J. Neurosci., 15: 315-328.
PubMedDirect Link - Balasko, M., Z. Szelenyi and M. Szekely, 1999. Central thermoregulatory effects of neuropeptide Y and orexin A in rats. Acta Physiol. Hung., 86: 219-222.
PubMed - De Jesus, L., S. Carvalho, M. Ribeiro, M. Schneider, S. Kim, J. Harney and P. Bianco, 2001. The type 2 iodothyronine deiodinase is essential for adaptive thermogenesis in brown adipose tissue. J. Clin. Invest., 108: 1379-1385.
Direct Link - Ehmke, H. and A. Just, 2003. The orexins: linking circulatory control with behavior. Am. J. Physiol. Regul. Integr. Comp. Physiol., 285: 519-521.
Direct Link - Harrison, F.A. and W.M. Leat, 1975. Digestion and absorption of lipid in non-ruminant and ruminant animals: A comparison. Proc. Nutr. Soc., 34: 203-210.
PubMed - Goncz, E., M.Z. Strowski, C. Grotzinger, K.W. Nowak and P. Kaczmarek et al., 2008. Orexin-A inhibits glucagon secretion and gene expression through a Foxo1-dependent pathway. Endocrinology, 149: 1618-1626.
PubMed - Hagan, J.J., R.A. Leslie, S. Patel, M.L. Evans and T.A. Wattam et al., 1999. Orexin A activates locus coeruleus cell firing and increases arousal in the rat. Proc. Natl. Acad. Sci., 96: 10911-10916.
Direct Link - Holl, R.W., P. Bucher, W. Sorgo, E. Heinze, J. Homoki and K.M. Debatin, 1999. Supression of growth hormone by oral glucose in the evaluation of tall stature. Horm. Res., 51: 20-24I.
CrossRef - Jones, D.N., J. Gartlon and F. Parker, 2001. Effects of centrally administered orexin-B and orexin-A: A role for orexin-1 receptors in orexin-B-induced hyperactivity. Psychopharmacology, 153: 210-218.
Direct Link - Lanni, A., M. Moreno, A. Lombardi, P. de Lange and F. Goglia, 2001. Control of energy metabolism by iodothyronines. J. Endocrinol. Invest., 24: 897-913.
Direct Link - Legradi, G., C.H. Emerson, R.S. Ahima, J.S. Flier and R.M. Lechan, 1997. Leptin prevents fasting-induced suppression of prothyrotropin-releasing hormone messenger ribonucleic acid in neurons of the hypothalamic paraventricular nucleus. Endocrinology, 138: 2569-2576.
CrossRefPubMedDirect Link - Legradi, G., C.H. Emerson, R.S. Ahima, W.M. Rand, J.S. Flier and R.M. Lechan, 1998. Arcuate nucleus ablation prevents fasting-induced suppression of ProTRH mRNA in the hypothalamic paraventricular nucleus. Neuroendocrinology, 68: 89-97.
Direct Link - Lopez, M., L.M. Seoane, S. Tovar, R. Nogueiras, C. Dieguez and R. Senaris, 2004. Orexin-A regulates growth hormone-releasing hormone mRNA content in a nucleus-specific manner and somatostatin mRNA content in a growth hormone-dependent fashion in the rat hypothalamus. Eur. J. Neurosci., 19: 2080-2088.
Direct Link - Manabe, T., Y. Okada, H. Sawai, H. Funahashi, M. Yamamoto, T. Hayakawa and T. Yoshimura, 2003. Effect of galanin on plasma glucose, insulin and pancreatic glucagon in dogs. J. Int. Med. Res., 31: 126-132.
Direct Link - Meghan, M. and K. Samson, 2003. The other side of the orexins: Endocrine and metabolic actions. Am. J. Physiol. Endocrinol. Meta., 284: 13-17.
Direct Link - Nowak, K.W., M.Z. Strowski, M.M. Switonska, P. Kaczmarek and V. Singh et al., 2005. Evidence that orexins A and B stimulate insulin secretion from rat pancreatic islets via both receptor subtypes. Int. J. Mol. Meta., 15: 969-972.
Direct Link - Mitsuma, T., Y. Hirooka, Y. Mori, M. Kayama and K. Adachi et al., 1999. Effects of orexin A on thyrotropin-releasing hormone and thyrotropin secretion in rats. Horm. Metab. Res., 31: 606-609.
PubMed - Overeem, S., S.W. Kok, G.J. Lammers, A.A. Vein and M. Frolich et al., 2003. Somatotropic axis in hypocretin-deficient narcoleptic humans: Altered circadian distribution of GH-secretory events. Am. J. Physiol. Endocrinol. Metab., 284: 641-647.
CrossRefDirect Link - Quedraogo, R., R. Naslaud and A. Kirshgesner, 2003. Glucose regulate the release of orexin from the pancreas. Diabetes, 52: 111-117.
Direct Link - Russell, S.H., C.J. Small and C.L. Dakin, 2001. The central effects of orexin-A in the hypothalamic pituitary adrenal axis in vivo and in vitro in male rats. J. Neuroendocrinol., 13: 561-566.
Direct Link - Russell, S.H., C.J. Small, D. Sunter, I. Morgan, C.L. Dakin, M.A. Cohen and S.R. Bloom, 2002. Chronic intraparaventricular nuclear administration of orexin A in male rats does not alter thyroid axis or uncoupling protein-1 in brown adipose tissue. Regul. Peptides, 104: 61-68.
Direct Link - Towhidi, A., H. Khazali and M. Zhandi, 2007. Leptin Is a metabolic signal for GnRH-LH/FSH axis in feed-restricted ewes. Asian-Aust. J. Anim. Sci., 20: 1039-1048.
Direct Link - Van Haasteren, G.A.C., E. Linkels, W. Klootwijk, H. Van-Toor and J.M.M. Rondeel et al., 1995. Starvation-induced changes in the hypothalamic content of prothyrotrophin-releasing hormone (proTRH) mRNA and the hypothalamic release of proTRH-derived peptides: Role of the adrenal gland. J. Endocrinol., 145: 143-153.
CrossRefDirect Link - Samson, W.K. and M.M. Taylor, 2001. Hypocretin/orexin suppresses corticotroph responsiveness in vitro. Am. J. Physiol. Regul. Integr. Comp. Physiol., 281: 1140-1145.
Direct Link








