Research Article
Drinking Water Denitrification in a Packed Bed Anoxic Reactor: Effect of Carbon Source and Reactor Depth
Department of Civil Engineering, Faculty of Engineering, King Abdul Aziz University, P.O. Box 80204, Jeddah 21589, Saudi Arabia
Nitrate contaminated groundwater is becoming serious problem in many parts of the world. This contamination mainly results from excessive fertilization and uncontrolled discharge of treated wastewater (Gayle et al., 1989; Vasiliadou et al., 2009). High levels of nitrate in drinking water can cause methemogabinemia (blue baby disease) in infants. Nitrate also produces carcinogenic nitrosamines (Sarina and Rheinheimer, 2004; Liu et al., 2009).
To protect consumers from the adverse effect of nitrate contaminated drinking water, many organizations have set standards to regulate nitrate concentration. The drinking water standard set by the European Economic Community (EEC) is 50 mg L-1 as nitrate (11.3 mg L-1 -N) and the standard set by U.S Environmental Projection Agency (EPA) is 44 mg L-1 as nitrate (10 mg L-1 - N).
Several methods are available for the removal of nitrate from water (Aslan and Turkman, 2003; Lin et al., 2008). Dilution of high nitrate groundwater with low nitrate water is the main method to lower the nitrate in drinking water to the allowable limit. Nitrate removal from drinking water (denitrification) can be achieved through physical, chemical and biological processes. The physical and chemical processes include, ion exchange, reverse osmosis, chemical reduction, electrodialysis and distillation (Eisentraeger et al., 2001; McAdam and Judd, 2007). The main advantage of the physical and chemical processes is their simplicity and reliability, while, the disadvantage of these processes is the production of large volumes of brine with high nitrate, sulfate and chloride concentrations. Brine disposal can be very difficult and cause financial and environmental problems (Soares, 2002; Fabbricino and Petta, 2007).
Biological removal of nitrate appears to be an effective and promising method. However, the available studies have reported a wide range of denitrification performance depending on the carbon source and reactor type (Gayle et al., 1989; Gomez et al., 2000; Lee et al., 2001; Aslan and Turkman, 2003). The present study was conducted to evaluate the performance of an anoxic upflow packed-bed reactor to remove nitrate from drinking water. The effect of different types of substrate on the performance of the process and the optimum depth of the reactor was also evaluated. This study will provide useful information regarding some of the specific requirements for designing and building large treatment plants.
Location and details of experimental setup: This research project was conducted from June, 2008 to May, 2009 at the King Abdul Aziz University campus, Jeddah, Saudi Arabia.
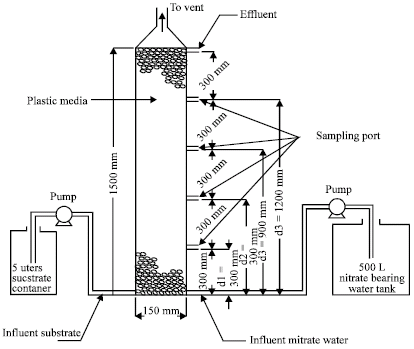 | |
| Fig. 1: | Experimental setup |
A laboratory scale anoxic upflow packed-bed reactor was used. The reactor was made of PVC column with an inner diameter of 150 mm and a height of 1.50 m (Fig. 1). The lower end of the column was completely sealed using 6 mm thick plexiglass sheet. An inverted cone was used at the upper end of the column to support the media of the column and to provide the necessary ventilation.
The reactor was packed with synthetic media (25 mm cylindrical rings) as the bacterial support matrices. Sampling ports were installed along the reactor at 300 mm intervals. Two inlets were located at the lower end of the reactor; one was used for the feed water and the other inlet was used for the carbon source. The sampling ports and the inlets were equipped with control valves.
Two positive displacement peristaltic pumps (Cole-Parmer pumps with Master-Flex pump heads) were used to operate the experimental system. One pump was used to transfer the nitrate-bearing water to the reactor and the second pump was used to transfer the substrate (the carbon source) to the reactor.
Silicon tubes were utilized to facilitate flows during the experimental work. All tubes were always replaced by clean ones to avoid any leakage or contamination within the system. The system was washed, cleaned and tested before starting the experiments and checked for leakage.
Nitrate bearing water: The nitrate bearing water was prepared by adding a sufficient amount of sodium nitrate and other nutrient to tape water to obtain the desired nitrate loading (200 mg L-1 as nitrate) in a 500 L fiberglass tank. About, 1 mg L-1 of phosphorus was added to the nitrate bearing water by adding potassium diphosphate solution, to ensure that phosphorus would not be a limiting nutrient for bacterial growth. The dissolved oxygen concentration in the nitrate bearing water was reduced by bubbling nitrogen gas in the nitrate bearing water tank, to ensure anaerobic condition in the reactor.
Substrate: Four types of organic compounds (namely; ethanol, acetic acid, dextrose and formic acid) were used as substrates and as sources of carbon in the experimental program. The substrate solution was made by adding the required volume of the organic compound to tap water in a 5 L glass container, they are:
Ethanol: The ethanol solution was prepared by adding 32.5 mL of ethanol to 5 L of tap water. This solution was pumped at a rate of 0.6 mL min-1 to maintain the ratio of carbon to nitrogen (C:N) of 0.87 as per the stoichiometric requirement.
Acetic acid: The acetic acid solution was prepared by adding 34 mL of acetic acid to 5 L of tap water. This solution was pumped at a rate of 0.6 mL min-1 to maintain the ratio of carbon to nitrogen (C:N) of 2.24 as per the stoichiometric requirement:
Dextrose: The dextrose solution was prepared by adding 14.5 mg of dextrose to 5 L of tap water. This solution was pumped at a rate of 0.6 mL min-1 to maintain the ratio of dextrose to nitrate of 0.208 as per the stoichiometric requirement.
Formic acid: The formic acid solution was prepared by adding 66.67 mL of formic acid to 5 L of tap water. This solution was pumped at a rate of 0.6 mL min-1 to maintain the ratio of formic acid to nitrate of (2:1) as per the stoichiometric requirement.
Start-up and operation: The reactor was seeded using an activated sludge sample obtained from King Abdulaziz University Wastewater Treatment Plant. About 4 L of active biomass was collected and distributed over the surfaces of the filter media. After seeding, pumping the nitrate bearing water and the substrate solution were started.
Analytical methods: Samples were taken from the influent and effluent and analyzed for the nitrate, pH, Dissolved Oxygen (DO) and alkalinity. When steady state was reached, samples were taken from the influent, effluent and sampling ports at various levels (300, 600, 900 and 1200 mm) and analyzed for nitrate, nitrite, pH, Dissolved Oxygen (DO), alkalinity, Chemical Oxygen Demand (COD) and turbidity (Table 1) following the standard methods.
| Table 1: | Parameters analyzed |
 | |
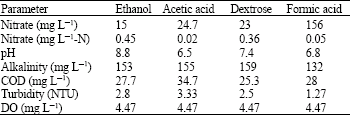
Denitrification performance: As shown Table 2, under the tested conditions the denitrification was the best in case of ethanol (average residual NO3¯ concentration of 15 mg L-1 with an influent concentration of around 200 mg L-1), while formic showed the worst performance (effluent nitrate concentration of 156 mg L-1). The denitrification performance in case of acetic acid and dextrose was more or less similar (effluent nitrate concentration of 24.7 and 23, respectively).
Except in case of formic acid, the steady state nitrate removal was achieved in general within 2 weeks or so. Despite several trials with different concentrations of formic acid (Fig. 2-7), a satisfactory denitrification rate could not be achieved. Formic acid may have been toxic to the microbial agent to some extent. Further study in this aspect would be interesting.
As shown in Fig. 2, in case of ethanol, the nitrate removal stabilized within 2 weeks. The average influent nitrate concentration was 203 mg L-1, while the effluent concentration was 15 mg L-1. In case of acetic acid, the average effluent nitrate concentration was 24.7 g L-1 against an average influent nitrate concentration of 206 mg L-1 (Fig. 3). The nitrate removal also stabilized within 2 weeks in this case. The time taken for stable nitrate removal was also very similar when dextrose was used as the carbon source.
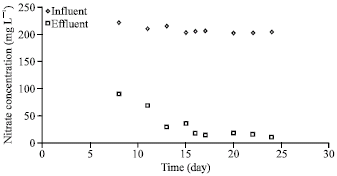 | |
| Fig. 2: | Denitrification performance (ethanol as carbon source) |
| Table 2: | Average change of different parameters during denitrification in case of different carbon sources |
 | |
| COD: Chemical oxygen demand; DO: Dissolved oxygen | |
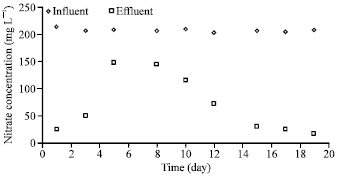 | |
| Fig. 3: | Denitrification performance (acetic acid as carbon source) |
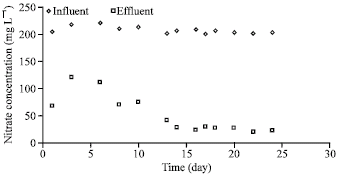 | |
| Fig. 4: | Denitrification performance (dextrose as carbon source) |
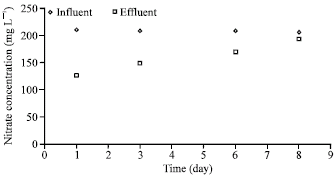 | |
| Fig. 5: | Denitrification performance (formic acid as carbon source; trial 1) |
In this case, the average effluent and influent nitrate concentrations were 23 and 202 mg L-1, respectively (Fig. 4). Figure 5-7 show the results of the trials using formic acid as the carbon source. In view of the very low removal of nitrate in case of the initial dose of 13.3 ml L-1 of formic acid (Fig. 5), the formic acid dose was stepwise reduced to 6, 3, 2.5, 2 and 1.5 ml L-1, respectively (Fig. 6, 7); however, nitrate removal did not improve significantly.
In the current study, while denitrification proceeded, accumulation of nitrite did not occur. The nitrite concentration in the effluent ranged from 0.002-0.45 only (Table 2). The fact that nitrate removal was achieved without encountering accumulation of nitrite makes the developed process very attractive.
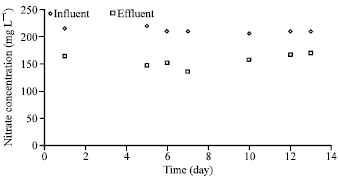 | |
| Fig. 6: | Denitrification performance (formic acid as carbon source, trial 2) |
 | |
| Fig. 7: | Denitrification performance (formic acid as carbon source, trial 3) |
Residual COD: Due to the addition of extra carbon sources, increased value of COD in the effluent is generally expected (Lee et al., 2001). Depending on the carbon source, the influent COD in the range of 3.7 to 4.7 increased up to the range of 25.3 to 34.7 mg L-1 (Table 2). The residual COD was the lowest in case of dextrose. Further improvement in reducing the residual COD may be required if direct use as drinking water is intended. This aspect is currently under study in our laboratory.
Change in pH: In case of ethanol, the average influent pH was 8 and the pH of the final effluent was 8.8 (Table 2). The corresponding values in case of acetic acid were 7.5 and 6.47, respectively, while in case of dextrose, the influent pH of 7.5 changed to 7.37 in the final effluent. Except in case of ethanol, the pH dropped from slightly to moderately after the treatment. Depending on the intended use, slight adjustment of the pH of the treated water may be required, especially in case of ethanol, for which the effluent is moderately alkaline.
Change in alkalinity: The alkalinity in the effluent was much higher irrespective of the carbon source used. The Table 3: Effect of depth on the removal of NO3 using ethanol influent alkalinity of the range of 30-33 mg L-1 rose up to the range of 125-147 mg L-1 (Table 2).
| Table 3: | Effect of depth on the removal of NO3 using ethanol |
 | |
| Table 4: | Effect of depth on the removal of NO3 using acetic acid |
 | |
The alkalinity was the highest in case of dextrose and the lowest in case of ethanol. Increase in alkalinity is inevitable due to the biological denitrification process (Gomez et al., 2000; Sarina and Rheinheimer, 2004) and further adjustments may be required depending on the intended use of the produced water.
Change in turbidity: The influent turbidity in all the cases was 1 NTU (Table 2). However, irrespective of the carbon source used, the effluent was four times more turbid than the influent. This may be attributed to the increase in alkalinity. This may be a concern from the aesthetic point of view. However, this can be taken care of by adjusting the pH, prior to the specific use of the treated water.
Change in dissolved oxygen: The average Dissolved Oxygen (DO) in the influent and effluent, irrespective of the carbon source used, were around 3.3 and 4.4, respectively (Table 2). Such DO level makes the produced water acceptable for high quality use (Fonseca et al., 2000).
Optimum depth of the reactor: Another practical aspect of this study, was the assessment of the reactor design. In order to reduce capital and operation cost a reactor as small as possible is practically expected (Aslan and Turkman, 2003). Accordingly, it is important to assess the optimum depth of the reactor. In this study, as expected, denitrification performance in case of each carbon source varied depending on the depth of the reactor (Table 3-6). In case of ethanol, while the nitrate concentration was 15 mg L-1 in the final effluent (1500 mm depth), the nitrate concentration from the outlet at 300 mm depth was 145 mg L-1. The corresponding values for acetic acid were 25 and 120 mg L-1, respectively. Similar trend was observed in case of dextrose and formic acid. The influence of reactor depth is hence evident.
| Table 5: | Effect of depth on the removal of NO3 using dextrose |
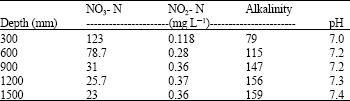 | |
| Table 6: | Effect of depth on the removal of NO3 using formic acid |
 | |
Like as denitrification performance, other related parameters also varied depending on the reactor depth (Table 2-6).
In our study, the denitrification varied depending on the carbon source utilized. The extent of denitrification achieved in this study is in accordance with the values reported in other biological processes (Gayle et al., 1989; Gomez et al., 2000; Lee et al., 2001; Aslan and Turkman, 2003).Under the tested conditions, the denitrification was the best in case of ethanol (average residual NO3¯ concentration of 15 mg L-1 with an influent concentration of around 200 mg L-1), while formic acid showed the worst performance (Table 7). A common concern in denitrification operations is the minimization of nitrite accumulation (Aslan and Turkman, 2003). This requires careful process control and more research regarding the nitrite problem is needed. The fact that nitrate removal was achieved without encountering accumulation of nitrite makes the developed process very attractive.
In addition to the denitrification, some other parameters were also monitored. According to our results, further improvement in reducing the residual COD was deemed required if direct use as drinking water is intended. This aspect is currently under study in our laboratory. Depending on the intended use, slight adjustment of the pH of the treated water may be also required, especially in case of ethanol, for which the effluent is moderately alkaline. Irrespective of the carbon source used, the effluent was four times more turbid than the influent. In order to remove solid particles from the effluent water, a sand filter unit may be placed after the denitrification reactor (Gayle et al., 1989; Eisentraeger et al., 2001).
Although ethanol showed the best denitrification performance, some of the other carbon sources showed slightly better performance in terms of the other monitored parameters.
| Table 7: | Summaries of the steady state results for the four different carobon sources |
 | |
| COD: Chemical oxygen demand; DO: Dissolved oxygen | |
For instance, the residual COD was the lowest in case of dextrose. On the other hand, despite achieving the best denitrification, the effluent pH in case of ethanol showed larger swings from neutrality. Nevertheless, from the point of denitrification, the best performance of ethanol is evident.
The main objective of this study was to investigate the effect of various carbon sources namely ethanol, acetic acid, dextrose and formic acid on the performance of anoxic upflow packed-bed reactor to remove the nitrate from drinking water. The study further investigates the degree of removal of nitrate at different depths of reactors for determining the optimum depth.
In this study, except in case of formic acid, the treated water satisfied both the EEC and EPA drinking water quality standard in terms of nitrate. Under the tested conditions, the denitrification was the best when ethanol was as the supplementary carbon source. Unlike as reported in other available studies, in the current study, accumulation of nitrite did not occur, which makes the developed process a very attractive one. Denitrification performance in case of each carbon source varied depending on the depth of the reactor. With the applied reactor diameter, a depth of 1500 mm was found to be adequate to achieve the required degree of denitrification. This study will provide useful information regarding some of the specific requirements for designing and building large treatment plants.