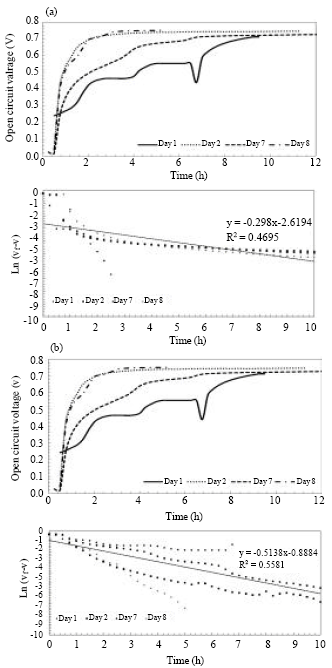
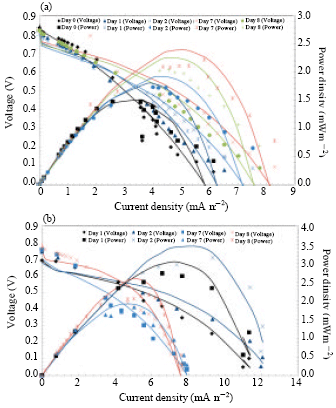
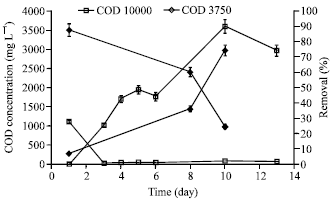
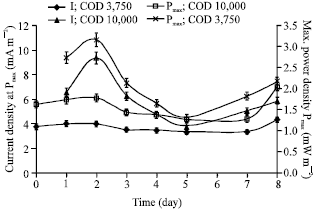
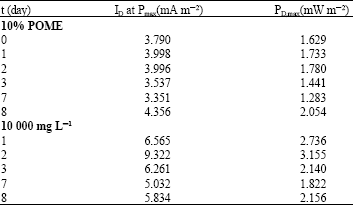
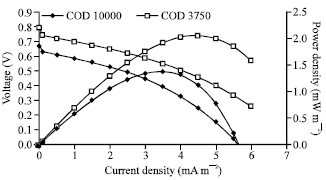
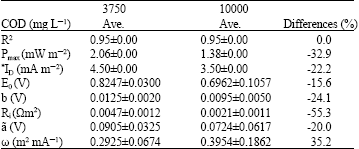
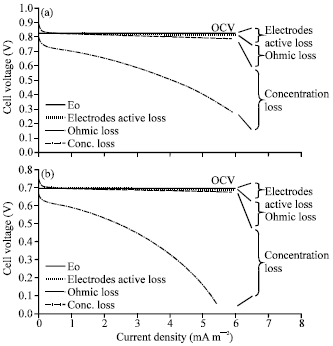
Research Article
Optimization of Electricity Generation and Palm Oil Mill Effluent (POME) Treatment from Microbial Fuel Cell
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Jamaliah Md. Jahim
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Wan Ramli Wan Daud
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Manal Ismail
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Nurina Anuar
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Siti Kartom Kamarudin
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia
Siti Norhana Shari
Fuel Cell Institute, Universiti Kebangsaan, 43600 Bangi, Selangor DE, Malaysia