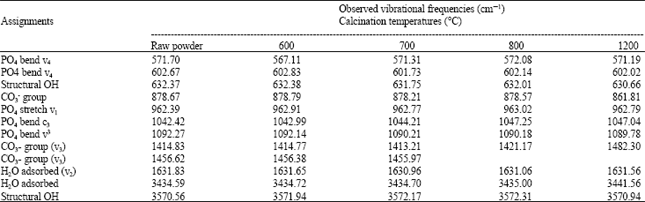
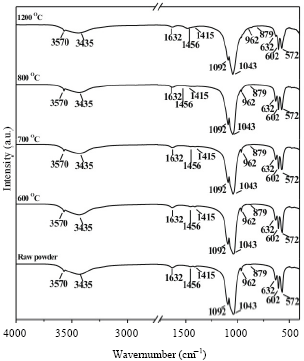
Research Article
Temperature Effect on Calcium Phosphate Synthesized from Chicken Eggshells and Ammonium Phosphate
Department of Physics, Faculty of Science, King Mongkut�s University of Technology Thonburi, Bangkok, 10140, Thailand
E. Hoonnivathana
Department of Physics, Faculty of Science, Kasetsart University, Bangkok, 10900, Thailand
P. Limsuwan
Department of Physics, Faculty of Science, King Mongkut�s University of Technology Thonburi, Bangkok, 10140, Thailand
K. Naemchanthara
Department of Physics, Faculty of Science, King Mongkut�s University of Technology Thonburi, Bangkok, 10140, Thailand