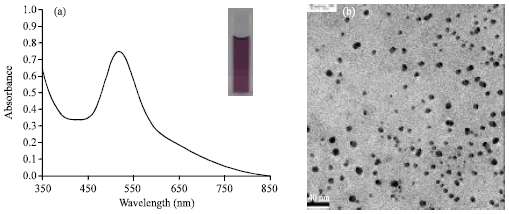
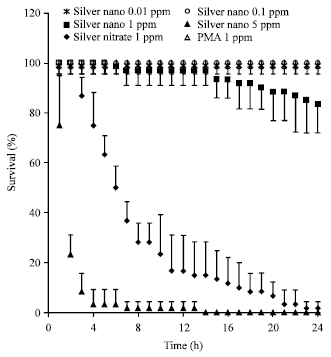
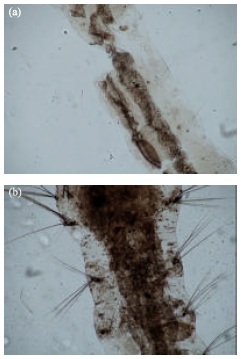
Short Communication
UV Irradiation-induced Silver Nanoparticles as Mosquito Larvicides
Pharmaceutical Technology (International Program), Department of Pharmaceutics and Industrial Pharmacy, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Bangkok, 10330, Thailand
C. Homklinchan
International School of Engineering, Faculty of Engineering, Chulalongkorn University, Bangkok, 10330, Thailand
R. Larpudomlert
International School of Engineering, Faculty of Engineering, Chulalongkorn University, Bangkok, 10330, Thailand
W. Warisnoicharoen
Department of Food and Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Bangkok, 10330, Thailand
A. Sereemaspun
Department of Anatomy, Faculty of Medicine, Chulalongkorn University, Bangkok, 10330, Thailand
S.T. Dubas
Metallurgy and Materials Science Research Institute, Chulalongkorn University, Bangkok, 10330, Thailand