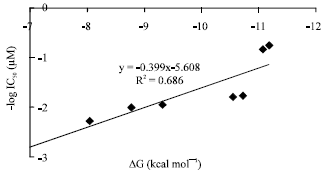
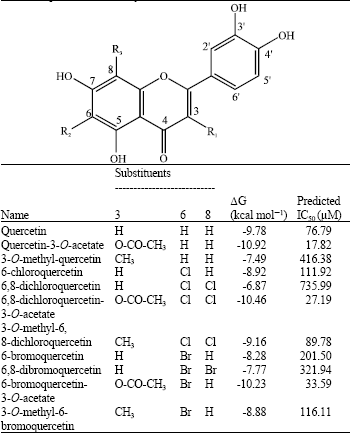
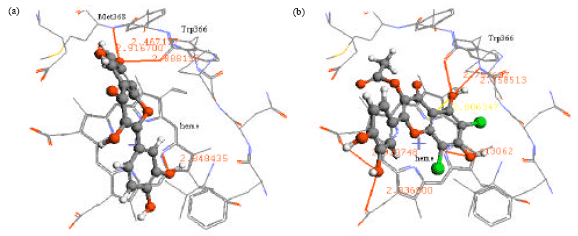
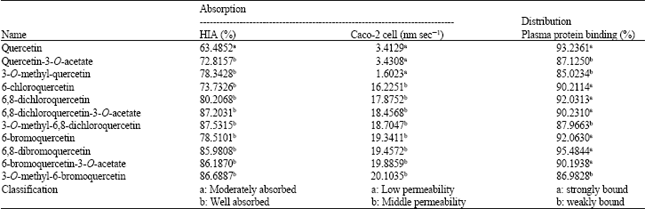
Research Article
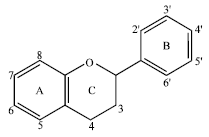
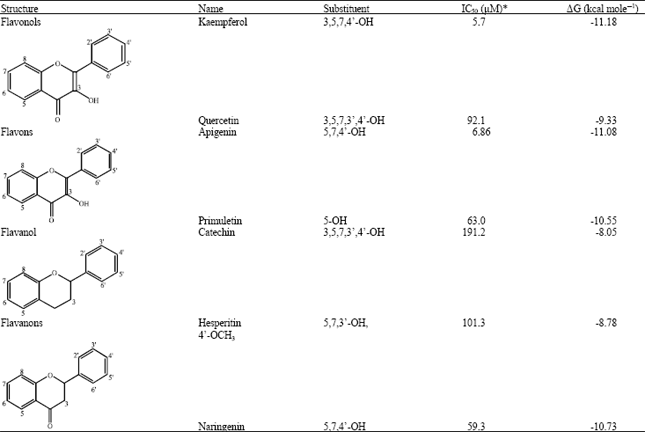
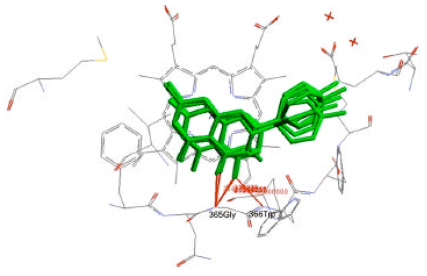
Docking Study of Quercetin Derivatives on Inducible Nitric Oxide Synthase and Prediction of their Absorption and Distribution Properties
School of Pharmacy, Institut Teknologi Bandung, Jalan Ganesha 10, Bandung, 40132, Indonesia
R. Herowati
School of Pharmacy, Institut Teknologi Bandung, Jalan Ganesha 10, Bandung, 40132, Indonesia
T. Gusdinar
School of Pharmacy, Institut Teknologi Bandung, Jalan Ganesha 10, Bandung, 40132, Indonesia