Research Article
Characterization of Tetracycline-loaded Thai Silk Fibroin/Gelatin Blend Films
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham 44150, Thailand
The blending method is a great advantage of materials since it can be tailored those of their properties by combining component and changing composition ratios of polymers. Generally, the blending process must be done in a fluid condition. Therefore, the dissolution of the polymers is also considered as an important step (Marsano et al., 2008). Moreover, natural homopolymer demands are not sufficient for biomaterial products (Fan et al., 2008). Recently, biopolymers such as: chitosan (She et al., 2008), gelatin (Martucci and Ruseckaite, 2009), cellulose and silk fibroin (Marsano et al., 2008) have been extensively studied due to their acted as potential environmentally friendly and biodegradability (Martucci and Ruseckaite, 2009).
Silk is a fibrous polymer that is produced by some Lepidoptera larvae including Bombyx mori (B. mori) silkworm (Altman et al., 2003). Each silk fiber consists of two types of proteins; fibroin and sericin. The silk fibroin (SF) is one of the candidate materials for biomedical applications. Recently, SF has been applied in various fields including cosmetics, medical materials and food additives (Min et al., 2004). Moreover, SF can be performed in many types depend on the applications. The SF film is one kind of regenerated form which was widely used in many fields. However, it often requires chemical treatments in order to enhance their stability and mechanical properties (Tasukada et al., 1995).
Gelatin (G), derivatives of collagen, is an edible and biodegradable polymer (Martucci and Ruseckaite, 2009). It shows excellent oxygen barrier as well as relative humidity and good mechanical properties (Jongjareonrak et al., 2006). All most reports, gelatin was performed in film form. The gelatin films tend to be brittle and moisture sensitive (Hernandez-Munoz et al., 2004). It has been used in pharmaceutical and medical fields (Rhim et al., 1998), drug delivery (Mandal et al., 2009) and wound dressings (Choi et al., 1999). The gelatin can be used alone or as a blend forms (Huang et al., 2004).
Tetracyclines are broad-spectrum antibiotics. They have been used for curing of bacterial infections over 50 years (Fritz and Zuo, 2007; Kolesnikov et al., 1996). This study was aimed to prepare the Thai silk fibroin/gelatin blend films for containing tetracycline which used as hydrophilic model drug. The some properties of the films such as morphology, chemical structure and percent transparency were investigated.
This study was performed for 4 months from March 1, 2010 to July 1, 2010. The experiments were carried out on Department of Chemistry, Faculty of Science. The characterization of all films properties were done at the Central Instrument, Faculty of Science, Mahasarakham University, Thailand
Materials: The Bombyx mori (locally called Nangnoi) silk cocoons were kindly supplied from Silk Innovation Center (SIC) Mahasarakham University, Thailand. All experimental reagents were analytical grade. Tetracycline hydro chloride was purchased from Sigma-Aldrich (USA).
Methods
Silk fibroin preparation: The silk cocoons were cut into small pieces and then firstly with boiled 0.5% (w/v) Na2CO3 to exclude sericin or gum. The silk fibroin (SF) was obtained and then used as subjected for preparation of SF solution.
Silk Fibroin (SF) solution: The pure SF was dissolved using tertiary solvent system of CaCl2-Ethanol-H2O (1:2:8 by mole), with magnetic stirred at 90-95oC for 2-3 h to obtain SF solution. The SF solution was then dialyzed using dialysis bag (MC = 7 kDa) against distilled water for 3 days. The obtained solution was calculated percent weight and kept for preparing SF films.
Gelatin (G) solution: The G solution was prepared at concentration of 1% wt. by weighing G powder for 1 g and then added distilled water up to 100 mL. The mixture was stirred for 30 min at room temperature until the G powder completely dissolved.
SF/G blend films preparation: The several of SF/G blend films were prepared by mixing SF/G solution at various ratios of 1:0, 3:1, 1:1, 1:3 and 0:1 by volume, respectively. The total volume of 20 mL in each composition was mixed with 0.04% w/v tetracycline and then poured on the 5 cm polystyrene plates. The plates were left at 40°C in an oven for 3 days to obtain films.
Characterization of blend films: The morphology of SF/G blend films with tetracycline were observed under scanning electron microscopy (SEM) (JEOL JSM-6460LV, Japan). The films were firstly cut into small pieces and then mounted on the stub with double side carbon tapes. The stubs were then sputter coated with gold to enhance surface conductivity.
The secondary structures of the films were analyzed by using Fourier transform infrared (FTIR) spectroscopy (Perkin Elmer-Spectrum Gx, USA) in the spectral region of ~4000-400 cm-1 at 4 cm-1 spectral resolution and 32 scans.
Thermal properties were measured using TA instruments, SDT Q600 (Luken’s drive, New Castle, DE). The films weight of 8-10 mg were prepared and loaded in a platinum crucible. The samples were non-isothermal heated from 50 to 700°C at a heating rate of 20°C min-1. The TGA was carried out in nitrogen with the flow rate of 100 mL min-1. The TG and heat flow were recorded with TA Instrument’s Q series explorer software. The analysis of the data were done using TA Instrument’s Universal Analysis 2000 software (version 3.3B).
Percent transparency of the films was studied by UV-Vis spectrophotometer (Geneq-Genesys 20) with wavelength at 660 nm.
Morphology: As shown in Fig. 1a-e, the native SF showed rougher of surface area than those of other blend films. The smooth surfaces of blend films gradually increased when the SF component decreased. With cross section, the native SF showed smooth surface with homogeneous phase through out the film. SF/G at 2:1 ratio, separate phase of the film surface was found. However, the separate phase was decreased when the SF content decreased. In addition, smooth surfaces can be obtained when blended SF and G at 1:2 ratio.
Chemical structure: The chemical structures of all SF/G blend films were analyzed using FTIR. The results showed strong region for 3 zones: amide I (1700-1600 cm-1), amide II (1600-1500 cm-1) and amide III (1250-1150 cm-1) (Fig. 2). FTIR results indicated that SF/G blend films composed of the mixture characteristics both SF and G, compared to native films. At amide I region, native G film showed the absorption peak at 1632 cm-1 with shoulder peak at 1661 cm-1 whereas, SF showed the absorption peaks at 1632, 1661 and 1697 cm-1 in similar intensity. The absorption peaks from 1500-1300 cm-1 occurred slightly different profile depended on the components of SF and G. In contrast, the amide III regions showed similar peaks in all of blend films.
Thermal properties: Thermogravimetric (TG) curves of SF/G blend films were shown in Fig. 3. G native films decomposed its weight in lower than other films until 300°C. After that, the blend films rapidly decomposed in maximum rate in range from 300-350°C. The native G films remain its weight the lowest at 400°C about 33% as well as 600°C about 22%. The weight of blend films gradually decreased when the content of G increased. The results of differential thermogravimetric (DTG) curve showed slight difference depended on the components of SF and G (Fig. 4). The maximum decomposition temperatures of each blend films were 316°C (3:0 ratio), 321°C (2:1 ratio), 316 and 334°C (1:1 ratio), 318°C (1:2 ratio) and 334°C (2:1 ratio) (Table 1).
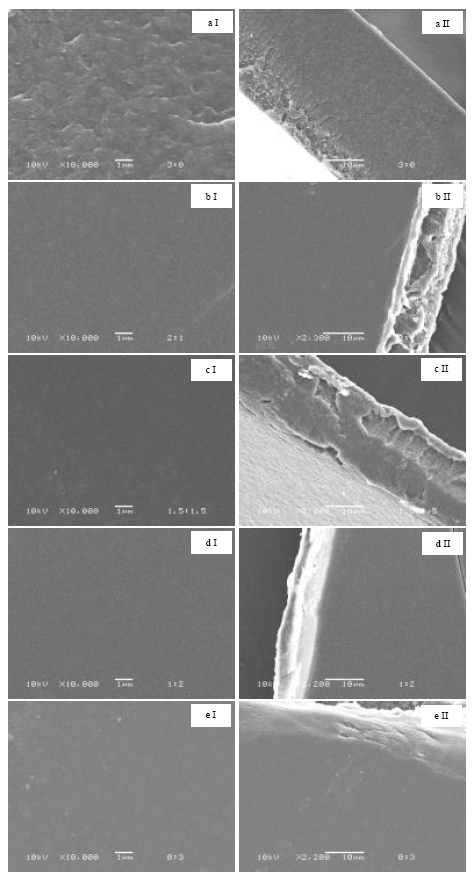 | |
| Fig. 1: | SEM micrographs of SF/G blend films with different ratios: (a) 3:0, (b) 2:1, (c) 1:1, (d) 1:2 and (e) 0:3. Note surfaces and cross section were presented in column I and column II. Most contained 0.04% tetracycline |
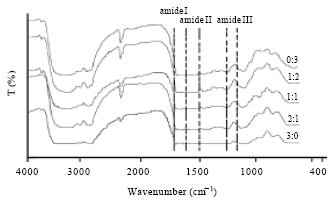 | |
| Fig. 2: | FTIR spectra showed amide regions of SF/G blend films with different ratios |
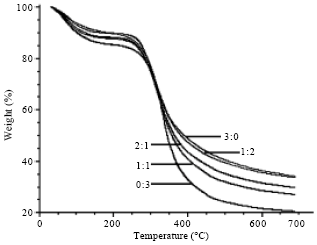 | |
| Fig. 3: | TG curves of SF/G blend films with different ratios |
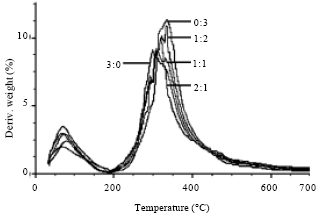 | |
| Fig. 4: | DTG curves of SF/G blend films with different ratios |
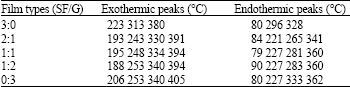
The native SF film showed one peak of endothermic at about 300°C while native G film showed endothermic peaks at least 3 points at about 227, 330 and 360°C. The exothermic peaks of both native films showed broad peaks at about 200 and 400°C.
| Table 1: | Maximum decomposition temperatures of SF/G blend films |
 | |
| Table 2: | Multiple Exo/Endo-thermic peaks of SF/G blend films with different ratios |
 | |
| Table 3: | Percent transparency of SF/G blend films with different ratios. (n = 4) |
 | |
Moreover, all of blend films showed slightly different of endo/exo-thermic peaks depending on the components of SF and G which were listed in Table 2.
Percent transparency: Table 3 showed the native SF and G films without tetracycline have higher percent transparency than all of films that composed of tetracycline. The percent transparency gradually decreased when the content of G increased. The lowest of percent transparency obtained in the native G film contained tetracycline at 37.50±1.80%.
The SF and G are biomaterials which were wildly used especially biomedical applications according to their suitable properties including biocompatibility, high and mechanical strength (Jin et al., 2004). Both are protein, however, they are different types of amino acids composition. This point might be influenced to differences of morphology, chemical structures, percent transparency or even interaction with drug. On the other hand, drug loading on the material used for control release was highly increased. Tetracycline is an antibacterial which was applied in many fields (Enomfon-Akpan and Umoh, 2004).
With SEM micrographs, the native SF showed rougher surface than other films. It is suggested that SF composed of large or various molecules which affected on the interaction together. The films all display homogeneous structure, indicated a high level of miscibility of the blend (Yang et al., 2000). However, the obviously homogeneous surface was adjusted by decreasing of SF contents. This result indicated that G helps to improve both charge and bond formation of the blend films (Okhawilai et al., 2010). Conformational structure is significantly influenced on protein properties (Lee et al., 2003). The chemical structures of protein were sensitively analyzed by FTIR in the regions of amide I (1700-1600 cm-1), amide II (1600-1500 cm-1) and amide III (1300-1200 cm-1) (Hino et al., 2003). Generally, the structures of SF and G are arranged in random coil and α-helix structures, respectively (Songchotikunpun et al., 2008). The FTIR results indicated that SF/G blend films showed individually characteristics of both SF and G with the co-existed of α-helix and β-sheet structures (Mandal et al., 2009; Tao et al., 2007). The results might be suggested that high number of hydrogen bonds have been formed between carbonyl groups and amino groups of protein. The obtained result showed as same as previously reported by our group (Wilaiwan et al., 2010). In different ratios of SF/G blend films, percent transparency was differed. The decreasing of percent transparency was obtained when the G content was increased. The results indicated that tetracycline (hydrophilic drug) could be interacted very well with G (polar material) and affected on the film transparency. This result concluded that percent transparency of the films should be influenced by material charge (Remunan-Lopez and Bodmeier, 1997).
Thermal properties analysis showed that native G film has higher strength than other films. However, it was rapidly decomposed after maximum decomposition temperatures. The blend films showed higher decomposition steps than that of native films. This might be from the blending between the characteristics of SF and G (Kweon et al., 2001).
Those of SF/G blend films loaded-tetracycline were prepared and investigated for their morphology, chemical structures, thermal properties and percent transparency. All of SF/G blend and G films have smooth surfaces, except native SF film. The surfaces of the blend films depended on the content of both SF and G components. Chemical structures indicated that SF and G could be interacted by hydrogen bonds formation. The component ratio of SF and G was a key point for thermal properties of the blend films. In conclusion, SF/G blend films properties could be adjusted and applied for loading hydrophilic substances.
The author’s great fully thank Faculty of Science, Division of Research Facilitation and Dissemination, Mahasarakham University and Center of Excellence for Innovation in Chemistry, Commission on Higher Education, Ministry of Education, Thailand for financial support of this study.