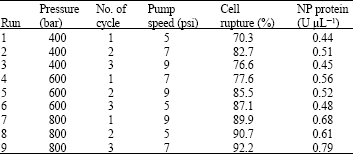
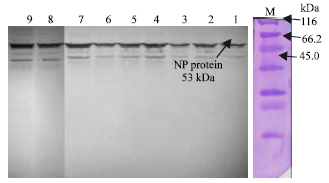
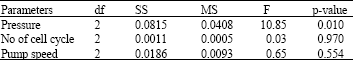
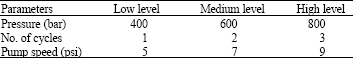Research Article
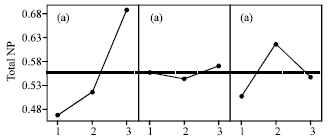
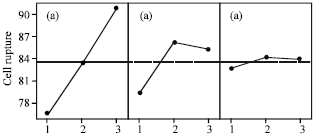
Cell Rupture of Recombinant Escherichia coli using High Pressure Homogenizer
Department of Biotechnology Engineering, Kulliyyah of Engineering, International Islamic University of Malaysia, Jalan Gombak, Kuala Lumpur, Malaysia
M. Mel
Department of Biotechnology Engineering, Kulliyyah of Engineering, International Islamic University of Malaysia, Jalan Gombak, Kuala Lumpur, Malaysia
S.N. Mohd-Abdullah
Department of Biotechnology Engineering, Kulliyyah of Engineering, International Islamic University of Malaysia, Jalan Gombak, Kuala Lumpur, Malaysia
K. Yusoff
Ministry of Science and Technology, Putrajaya, Selangor, Malaysia