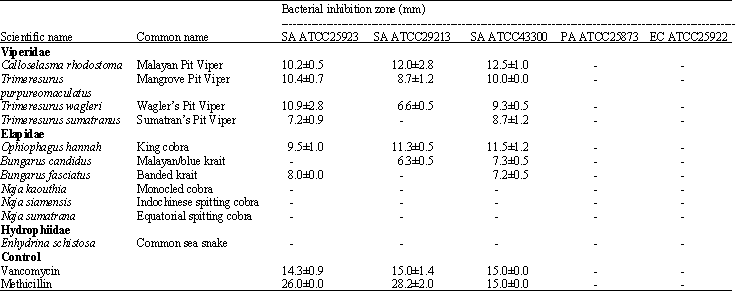
Research Article
Screening Antimicrobial Activity of Venoms from Snakes Commonly Found in Malaysia
Jeffrey Cheah School of Medicine and Health Sciences, Monash University Sunway Campus, Jalan Lagoon Selatan, Bandar Sunway, 46150, Selangor Darul Ehsan, Malaysia
J. Vejayan
Jeffrey Cheah School of Medicine and Health Sciences, Monash University Sunway Campus, Jalan Lagoon Selatan, Bandar Sunway, 46150, Selangor Darul Ehsan, Malaysia
K. Shanmugan
Jeffrey Cheah School of Medicine and Health Sciences, Monash University Sunway Campus, Jalan Lagoon Selatan, Bandar Sunway, 46150, Selangor Darul Ehsan, Malaysia
H. Ibrahim
Institute of Biological Sciences, University of Malaya, 50603 Kuala Lumpur, Malaysia