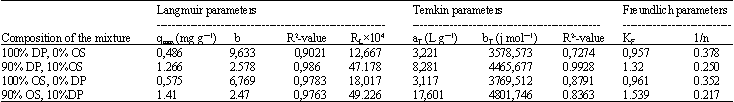
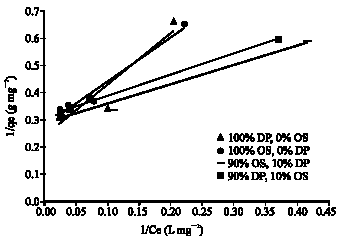
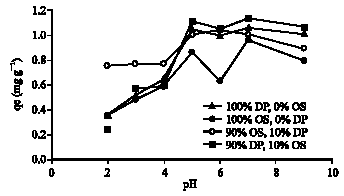
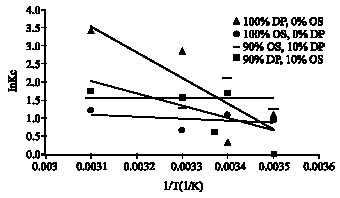
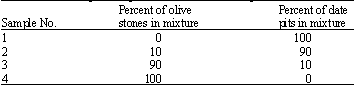Research Article
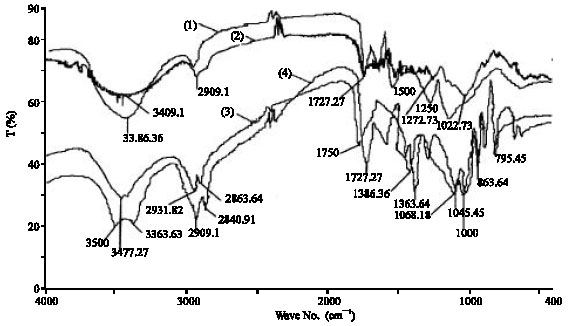
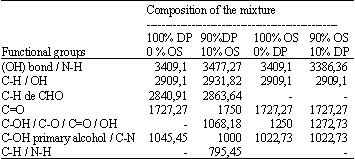
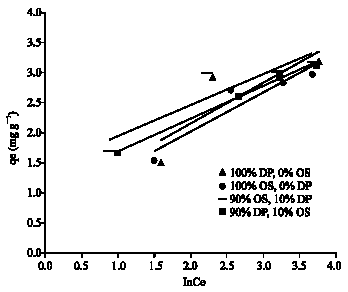
Adsorption of Cd(II) Ions from Aqueous Solution using Mixed Sorbents Prepared from Olive Stone and Date Pit
Laboratoire de Genie Alimentaire, Faculty of Engineer Science, University of Boumerdes, 35000, Boumerdes, Algeria
H. Aksas
Laboratoire de Genie Alimentaire, Faculty of Engineer Science, University of Boumerdes, 35000, Boumerdes, Algeria
S. Boughrara
Laboratoire de Genie Alimentaire, Faculty of Engineer Science, University of Boumerdes, 35000, Boumerdes, Algeria
K. Louhab
Laboratoire de Genie Alimentaire, Faculty of Engineer Science, University of Boumerdes, 35000, Boumerdes, Algeria