Research Article
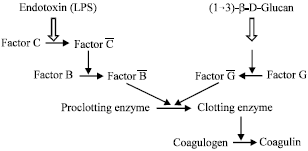
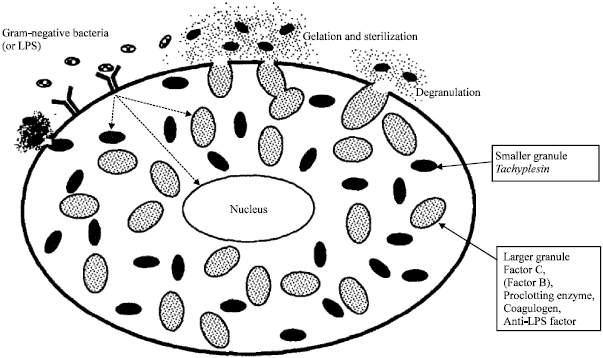
Mechanism in the Clot Formation of Horseshoe Crab Blood during Bacterial Endotoxin Invasion
Institute of Oceanography and Maritime Studies, International Islamic University Malaysia, Jalan Sultan Ahmad Shah, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
K.C.A. Jalal
Institute of Oceanography and Maritime Studies, International Islamic University Malaysia, Jalan Sultan Ahmad Shah, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
Y.B. Kamaruzzaman
Institute of Oceanography and Maritime Studies, International Islamic University Malaysia, Jalan Sultan Ahmad Shah, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
K. Zaleha
Institute of Aquatrop, Universiti Malaysia Terengganu, 21030 Kuala Terengganu, Terengganu, Malaysia










Amuzat Aliyu olalekan Reply
It is a useful and beneficial article to me