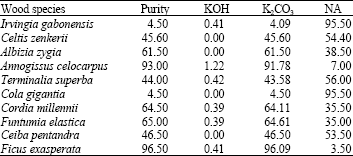
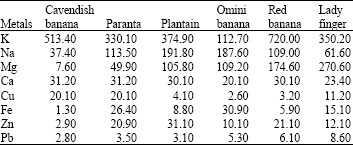
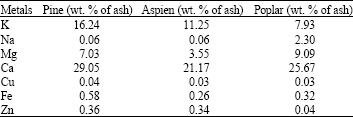
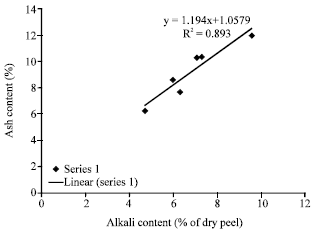
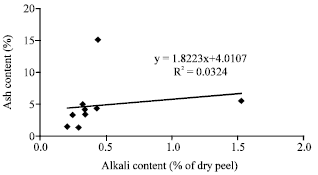
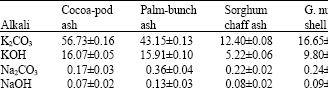Research Article
Evaluation of the Composition and Chemistry of Ash and Potash from Various Plant Materials-A Review
Department of Chemical Sciences, Bells University of Technology, Ota, Nigeria
K.T. Dauda
Department of Chemical Sciences, Bells University of Technology, Ota, Nigeria
D.O. Nwude
Department of Chemical Sciences, Bells University of Technology, Ota, Nigeria
A.A.A. Kayode
Department of Chemical Sciences, Bells University of Technology, Ota, Nigeria