Research Article
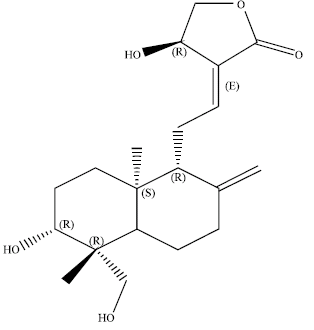
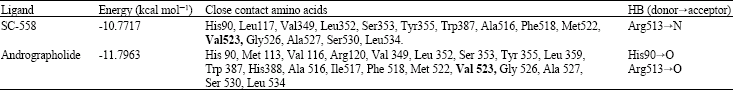
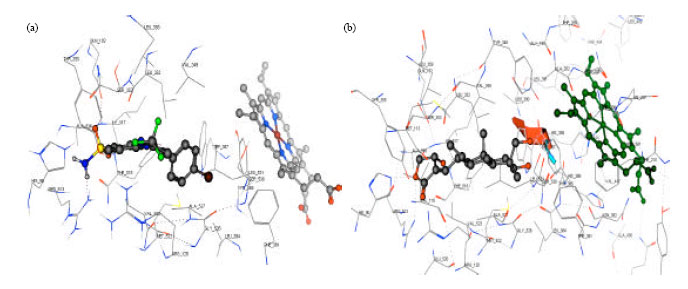
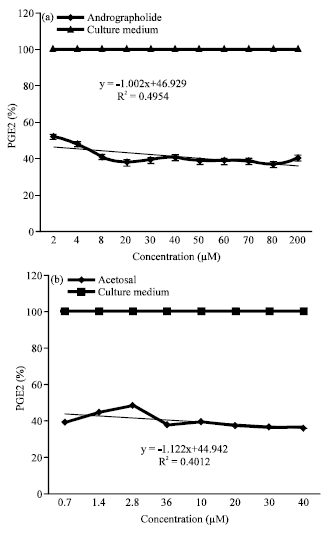
Andrographolide Inhibits COX-2 Expression in Human Fibroblast Cells Due to its Interaction with Arginine and Histidine in Cyclooxygenase Site
Faculty of Pharmacy, Universitas Padjadjaran,Jl. Raya Bandung-Sumedang Km. 21, Jatinangor, West Java, Indonesia
As `ari Nawawi
School of Pharmacy, Institut Teknologi Bandung,Jl. Ganesha 10 Bandung, West Java, Indonesia
Abdul Mutholib
Center for Radioisotope and Radiopharmaca, National Nuclear Agency, Serpong, Indonesia
Slamet Ibrahim
School of Pharmacy, Institut Teknologi Bandung,Jl. Ganesha 10 Bandung, West Java, Indonesia