Research Article
Neural Network and Genetic Algorithm Based Hybrid Model for Content Based Mammogram Image Retrieval
Mangalam College of Engineering, Kottayam, Kerala, India
P. Mythili
Division of Electronics, Cochin University of Science and Technology, Cochin, Kerala, India









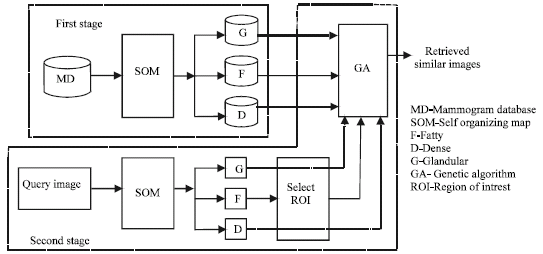
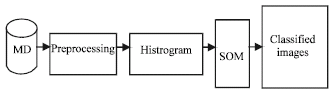
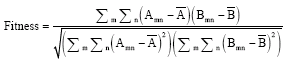
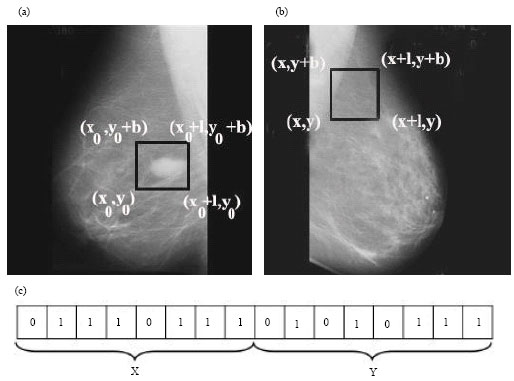


sudhakar Reply
can you send me the matlab code for this project
Justin Jose
May I Know Whom U r ? and For what purpose u need the Matlab Code
sudhakar Reply
i'm doing final project in matlab and from chennai