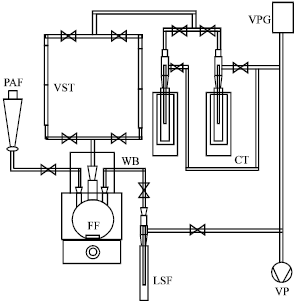
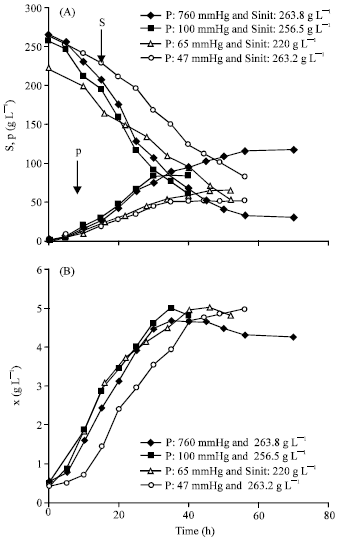
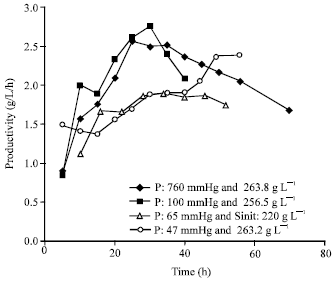
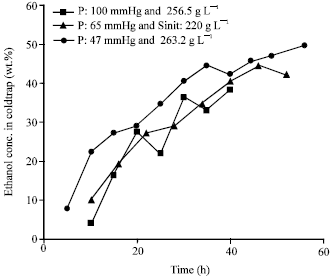
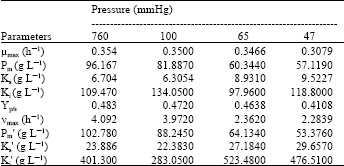
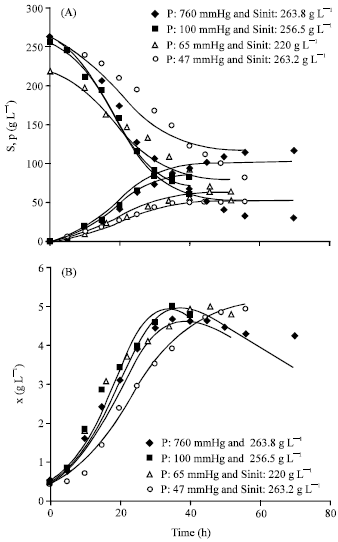
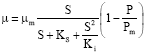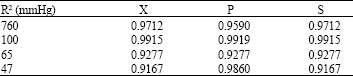Research Article
Effect of Vacuum Pressure on Ethanol Fermentation
Department of Chemical Engineering, College of Engineering, De La Salle University, Manila, 2401 Taft Avenue, Manila, Philippines
H. Kosuge
Department of Chemical Engineering, Tokyo Institute of Technology, 2-12-1, Ookayama, Meguro-Ku, Tokyo, 152-8552, Japan
J. Auresenia
Department of Chemical Engineering, College of Engineering, De La Salle University, Manila, 2401 Taft Avenue, Manila, Philippines
R. Tan
Department of Chemical Engineering, College of Engineering, De La Salle University, Manila, 2401 Taft Avenue, Manila, Philippines
Y. Brondial
Department of Chemical Engineering, College of Engineering, De La Salle University, Manila, 2401 Taft Avenue, Manila, Philippines