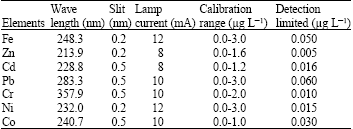
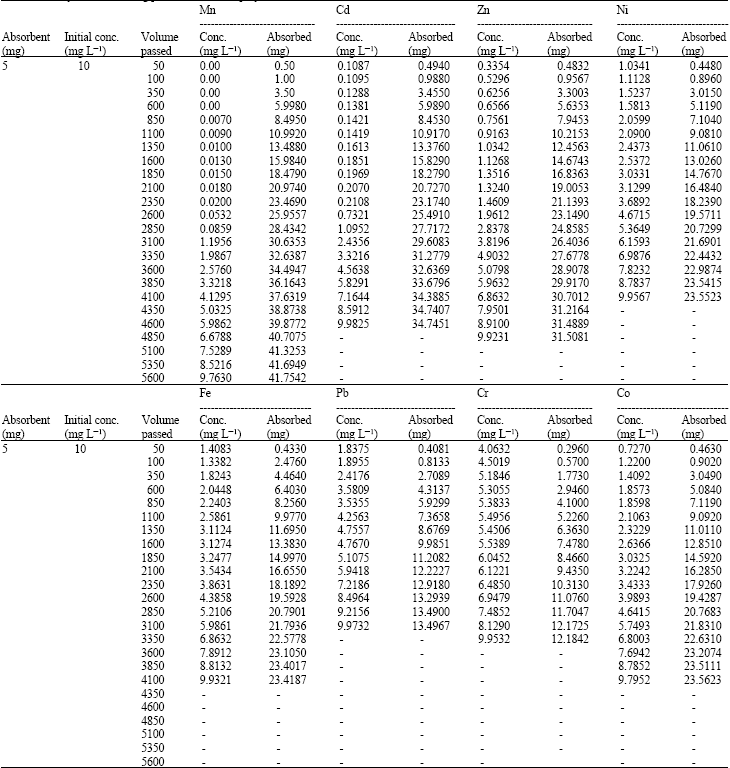
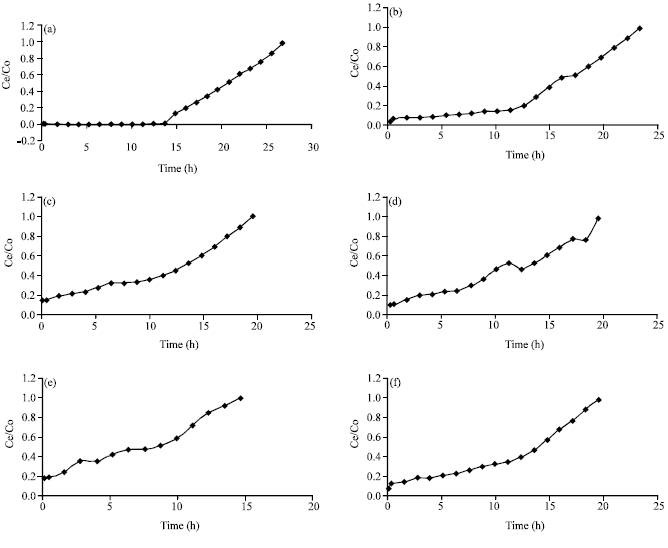
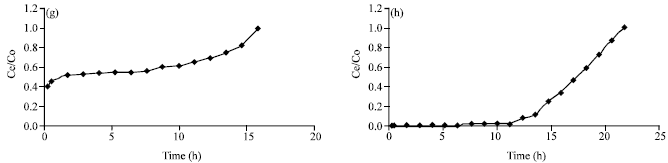
Research Article
Removal of Heavy Metal from Contaminated Water by Biopolymer Crab Shell Chitosan
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
M.A. Halim
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
S. Safiullah
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
M. Mamun Mollah
National Measurement Institute, Bertie Street, Port Melbourne, Victoria 3207, Australia
M.S. Azam
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
M.A. Goni
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
M. Kamal Hossain
Department of Chemistry, Jahangirnagar University, Savar, Dhaka, Bangladesh
M.M. Rana
Department of Biochemistry and Molecular Biology, Rajshahi University, Rajshahi, Bangladesh