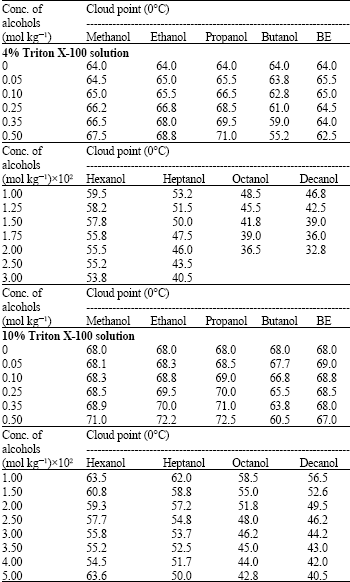
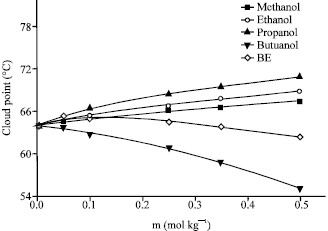
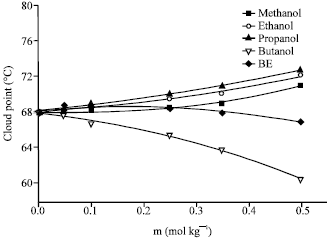
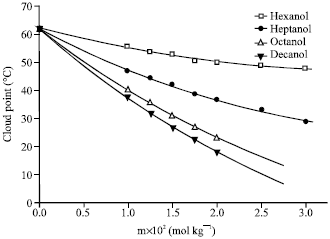
Research Article
Effect of Organic Additives on the Cloud Point of Triton X-100 Micelles
Department of Applied Chemistry and Chemical Technology, Islamic University, Kushtia 7003, Bangladesh
T. Parvin
Department of Applied Chemistry and Chemical Technology, Islamic University, Kushtia 7003, Bangladesh
T. Begum
Department of Chemistry, Chittagong University, Chittagong, Bangladesh