Research Article
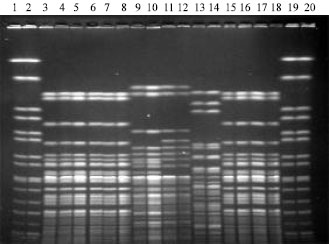
Use of Plastic Primer Container Covers as Effective Plug Molds for Pulsed-Field Gel Electrophoresis
Department of Molecular Medicine
Kah Heng See
Department of Molecular Medicine
S.D. Puthucheary
Department of Medical Microbiology, Faculty of Medicine, University of Malaya, 50603 Kuala Lumpur, Malaysia