Research Article
Measurement of Selected Enzymatic Activities in Solanum nigrum-Treated Biomphalaria arabica Snails
Department of Biochemistry, Science College, King Saud University, P.O. Box 22452, Zip Code 11495, Saudi Arabia
Schistosomiasis, also known as bilharziasis is a major parasitic disease of humans. It is thought to infect about 200 to 300 million people across Africa, South America, the Caribbean, the Middle East, China and Southeast Asia. The major causes of the disease are Schistosoma mansoni, Schistosoma japonicum and Schistosoma haematobium. Transmission of the schistosomes to humans is via contact with fresh water that contains the parasite`s intermediate snail host and that has been contaminated by urine or faecal material from infected individuals. As intermediate hosts, molluscs play a major role in the transmission of schistosomes; they are the sites of an intense multiplication of parasites. Thus, snail control strategies are considered a priority for the reduction of schistosomiasis (Lardans and Dissous, 1998). The high cost of synthetic molluscicides, used in the control of the intermediate snail hosts of schistosomiasis, has resulted in renewed interest in plant molluscicides. The prohibitive costs (time and financial) of random surveys for activity necessitated the development of an objective selection procedure. Species were ranked on cumulative scores for: (a) coincidence of the endemic areas of the plant, snail host and disease; (b) ethnomedicinal value which would provide greater incentive for cultivation and (c) molluscicidal activity (if known, a minimum LC90 of < or = 100 ppm) (Clark et al., 1997). A simple chemical will probably not be applicable for all situations; hence we must continue to investigate other control options and develop an integrated approach to schistosome-snail control. At present, the only viable molluscicide in terms of efficacy and complete evaluation is bayluscide but it is highly toxic to fish (LC50 0.03-0.23 mg L-1) (United States Environmental Protection Agency, 1999).
The development of new, specific and naturally occurring plant molluscicide was favoured by the World Health Organization (WHO). Many plant species have been evaluated as sources of naturally chemicals to control schistosome snails (Silva et al., 2007). The study of plants exhibiting snail toxicity has been encouraged with the aim of finding alternatives for use in the fight against snail vectors. The aim of studies on plant molluscicides is to complement methods for controlling snails acting as intermediate hosts of schistosomes. Attenuation of cercariae released from molluscicide-treated snails leading to reduced infectivity in mice could be achieved in concentrations below those exerting acute toxicity (i.e., sublethal concentrations) (El-Ansary et al., 2003; Abdel Kader et al., 2005).
Biological markers have been defined as xenobioticaly-induced variations in cellular or biochemical components in any biological system (Icen et al., 2005). Carbohydrate metabolizing enzymes (e.g., α-amylase), lipid degrading enzymes (e.g., lipase) and phosphatases (acid and alkaline phosphatases) were recorded among target enzymes which should be disturbed and may provide more accurate information on the molluscicide-induced stress on molluscs. Impairment of these enzymes could be effective in affecting the feeding and reproductive competence at the mollusc population level (fertility, fecundity and reproductive rate).The effect of sublethal concentrations of different synthetic and natural molluscicide agents on reproduction and biochemical aspects of schistosome snail hosts is recently encouraged (Abdel Kader et al., 2005). These information initiated our interest to measure ACP, ALP, amylase, lipase and glucose in tissue homogenates of B. arabica, the snail hosts to S. mansoni in Saudi Arabia. The present study was extended to evaluate the effect of sublethal concentration (LC25) of Solanum nigrum as a potent plant molluscicide on these biochemical parameters.
Snail collection and maintenance: Specimens of B. arabica were collected from a farm near Riyadh on June 2005. The snails were left in the lab for 45 days and were examined to be sure that they were free from parasitic infection. They were fed with lettuce leaves ad lib. A sample of the snails was randomly chosen and dissected.
Preparation of tissue homogenate: One gram of snail soft tissue was homogenized in 5 mL distilled water and then centrifuged at 3000 rpm, the supernatant was used for the biochemical analyses (Nabih et al., 1989).
Molluscicide-treatment: This was performed according to the toxicity study of S. nigrum plant on B. arabica snails previously done by El-Ansary and Al-Daihan (2007). Four groups 10 snails each were exposed to 3 ppm concentration of S. nigrum (LC25) dissolved in dechlorinated water in 1 L capacity tank. Snails were fed fresh lettuce leaves ad lib during the 24 h contact period. Dead snails were discarded and the remaining snails were used for the biochemical analysis. Untreated control groups were established.
Biochemical analysis Measurement of glucose: Glucose was measured according to the method of John and Turner (1975) using a diagnostic kit a product of United Diagnostic Industry (UDI), Riyadh, KSA.
Enzyme assays Measurement of phosphatases: ACP and ALP were measured according to the method of Bowers and McComb (1966), using a kinetic diagnostic kit, a product of United Diagnostic Industry (UDI), Riyadh, KSA.
Measurement of lipase: Lipase was measured according to the modified method of Vogel and Zieve (1963) turbidimetric method using a diagnostic kit a product of United Diagnostic Industry (UDI), Riyadh, KSA.
Measurement of amylase: Amylase was measured according to the method of Wallenfels et al. (1978) using a diagnostic kit a product of United Diagnostic Industry (UDI), Riyadh, KSA.
Statistical analysis: The statistical analysis of the data was carried out using Student t-test (GraphPad Prism computer program).
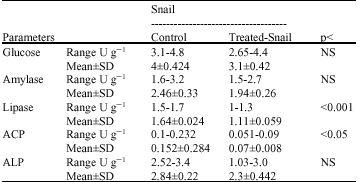
Table 1 shows the enzymatic activities of ALP, ACP, amylase, lipase and glucose in control and S. nigrum-treated B. arabica snails. Values are expressed as mean±SD of four different measurements. It can be easily seen that lipase was the most affected enzyme shows a highly significant lower activity in molluscicide-treated snails. ACP shows less significant reduced activity compared to control. On the other hand glucose, ALP and amylase were non-significantly affected. Figure 1 shows the percentage changes induced by the molluscicide in the measured parameters.
Ingested molluscicides are only partly transported down the digestive tract in the food mass and large proportion is quickly resorbed by the cells of the oesophagus (Triebskorn, 1995). After being released from these cells into the haemolymph, molluscicides molecules are distributed in the body quickly transported to peripheral tissues and organs.
In the present study, the molluscicidal activity of S. nigrum against B. arabica snails could be confirmed by the previous reports of Silva et al. (2005, 2006), Bekkouche et al. (2000) and Mkoji et al. (1989) who recorded a potent molluscicidal activity of this plant against B. glabrata, B. pfeifferi, Bulinus globosus and Lymnea natalensis, respectively.
| Table 1: | Enzymatic activities of ALP, ACP, amylase, lipase and glucose in control and S. nigrum-treated B. arabica snails |
 | |
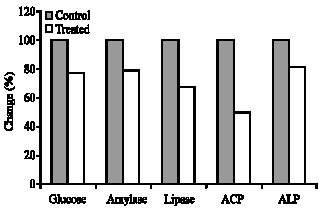 | |
| Fig. 1: | Percentage changes induced by the molluscicide in the ALP, ACP, amylase, lipase and glucose |
ACP and ALP have been shown to be influenced by molluscicide treatment, where the enzymes of this group can catalyses the breakdown of ester bonds in the orthophosphate esters under acidic and alkaline conditions, respectively. In B. glabrata, molluscan snail host to S. mansoni, ACP and ALP were detected histochemically among the enzymes that are important for the encapsulation reaction formed around schistosme sporocysts (McKerrow et al., 1985). A significant difference in the distribution and abundance of acid phosphatase as a lysosomal enzyme marker in blood cells (hemocytes) of schistosome-susceptible (PR albino M-line) and a resistant (10-R2) strain of B. glabrata during the course of infection with S. mansoni was recorded (Granath and Yoshino, 1983; Sasaki et al., 2005). They reported that, a significant increase of ACP 2 week PE, indicating a possible response to tissue damage resulting from migrating daughter sporocysts. On the other hand, alkaline phosphatase does not show any significant different in activity between normal and infected snails (Dardenne et al., 1979). In the present study, both enzymes were inhibited but only ACP showed statistically significant inhibition (p<0.008). This is in good agreement with the previous reports of Ahmed and Refaat (2004) who observed the inhibition of these enzymes in animals treated with naturally occurring molluscicides. Moreover, present results could be supported by the previous results of El-Ansary et al. (2000) and who recorded a potent molluscicidal activity of S. nigrum and recorded the impairment of hexokinase, glucose isomerase and pyruvate kinase in S. nigrum-treated B. alexandrina snails. In their study they recorded that LC10 concentrations of S. nigrum reduced considerably the infection rate of B. alexandrina with S. mansoni to be 34% compared to an infection rate of 80% in control, non-treated snails. Longer prepatent period and remarkable decrease in cercarial production. This confirms that sublethal concentration of S. nigrum is effective in disturbing the metabolism of the snail host to be less compatible to S. mansoni parasite (El-Ansary et al., 2000).
Nabih et al. (1998) reported that B. alexandrina and Bulinus truncatus as mo lluscan hosts to S. mansoni and S. haematobium, respectively have higher α-amylase activity and lower Km value to starch when compared to Lymnea truncatula and Physa acuta as non-target to schistosome parasites. They recorded that highly active amylase is of critical importance for the success of host-parasite relationship. Highly active amylase could easily provide the developing parasite with glucose as important nutrient needed by the developing intramolluscan parasite. In the present study, lower activity of α-amylase was observed in S. nigrum-treated snails, which could be easily correlated to the recorded lower glucose level. Importance of glucose for the development of schistosome larvae in B. arabica as a molluscan hosts to S. mansoni in Yemen was previously recorded (El-Sheikh and Nagi, 1991) who reported that Schistosome infection caused a marked decrease in tissue glycogen and protein of B. arabica and remarkable lower glucose content in the hemolymph of infected snails compared to control.
P31 NMR studies demonstrated that the levels of free phospholipids, particularly phosphatidylcholine, in the digestive gland of the snail, B. glabrata, were dramatically reduced when the host was infected with S. mansoni. It was speculated that absorption of host phosphatides may be an important source of membrane phospholipid precursors and fatty acids for developing sporocysts and cercariae (Thompson et al., 1991). Regarding the effect of molluscicide treatment on the enzymatic activity of lipase, it can easily be noticed that lipase was the most affected enzyme showing very high significant inhibition (p<0.0002). The remarkable inhibition of lipase in S. nigrum-treated snails could easily render the snail host physiologically unsuitable for the development of schistosome parasite since it is well known that schistosomes are unable to synthesize fatty acids de novo but absorbed it from their snail host tissues and haemolymph.
The obtained data could be supported by the previous reports of Ahmed and Ramzy (1997) who recorded that B. alexandrina snails exposed to sublethal concentration of S. nigrum shed significantly fewer cercariae than unexposed snails. Moreover it could clarify the previous study of El-Ansary et al. (2003) which reported that attenuated cercariae released from S. nigrum-metabolically disturbed snails has reduced pathogenicity to the mammalian hosts. Reduction of cercarial number released from S. nigrum treated snails (Ahmed and Ramzy, 1997) could be attributed to the significant lower ACP activity. Lower ACP activity could induce the cellular immune response of the host resulting in the killing of larger number of intramolluscan schistosome larvae, which lead to shedding of fewer cercariae. Moreover, lower lipase activity could lead to the production of cercariae with disturbed lipid profile which could be easily correlated to the lower pathogenicity to the mammalian hosts previously reported by El-Ansary et al. (2003). In view of the present results, S. nigrum could become an affordable and effective component of an integrated approach to schistosomiasis control in Saudi Arabia.
The authors are grateful for the Deanship of Scientific Research, King Saud University, Kingdom of Saudi Arabia for Supporting this work as an Applied National Research Project Grant DSR-AR-2 (35).
Prof.Dr. Ahmed Tarek Sharaf El-Din Reply
This paper is a master piece of science. But it needs some explanation in the discussion especially how these plant extract affect enzymes of snails (mode of action).