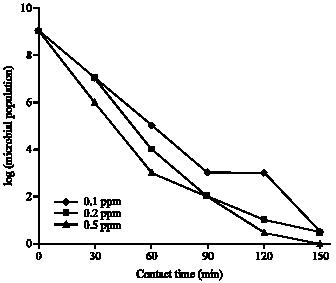
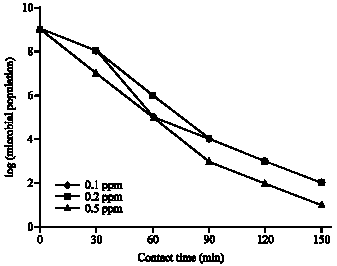
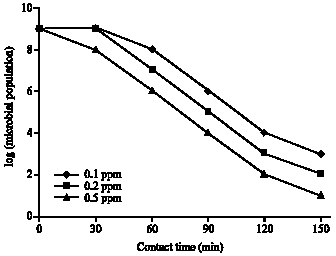
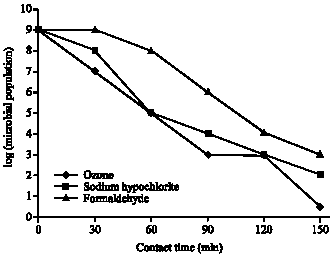
Research Article
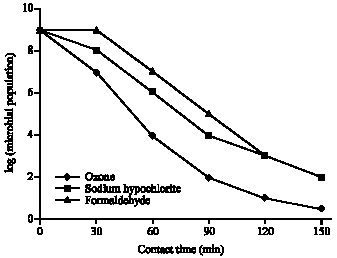
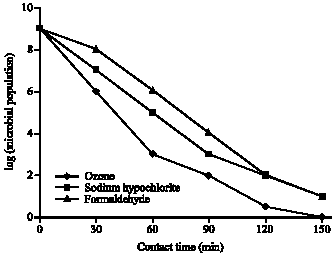
Biocidal Efficacy of Dissolved Ozone, Formaldehyde and Sodium Hypochlorite Against Total Planktonic Microorganisms in Produced Water
Department of Chemical Engineering, Rivers State University of Science and Technology, Port Harcourt, P.M.B. 5080, Port Harcourt, Nigeria
A. Rim-Rukeh
Department of Integrated Science, College of Education, P.M.B. 2090, Agbor, Delta State, Nigeria