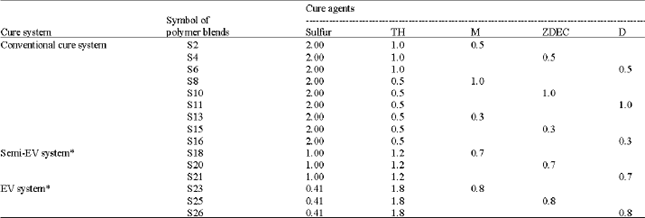
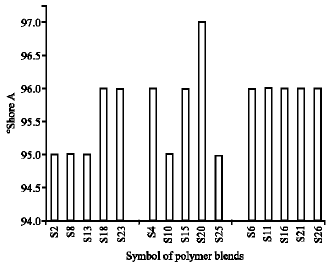
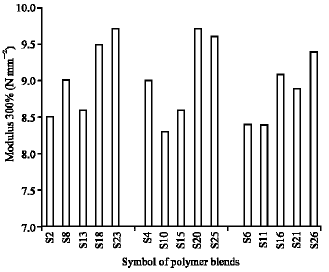
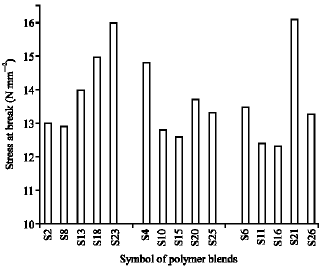
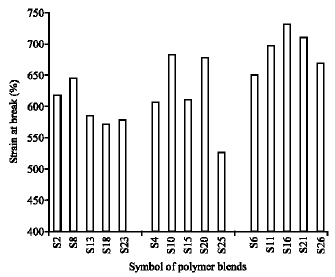
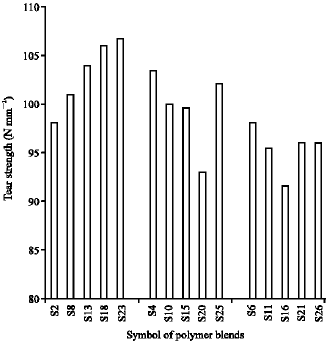
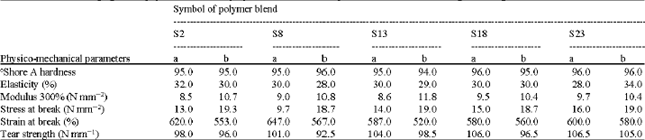
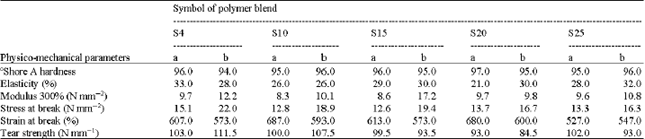
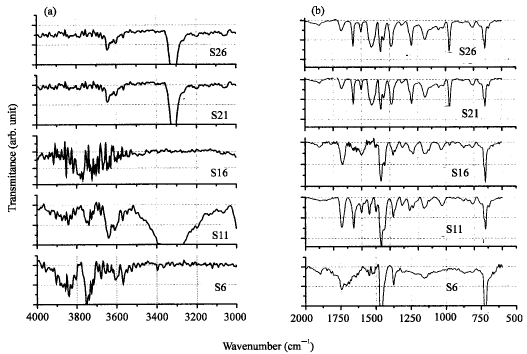
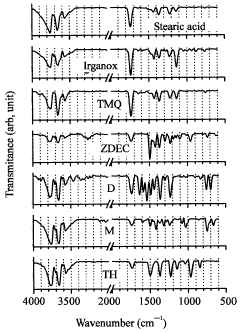
Research Article
EPDM-HDPE Blends with Different Cure Systems/Mechanical and Infra-Red Spectrometric Properties
Department of Analytical Chemistry, Faculty of Chemistry, University of Bucharest, 4-12 Regina Elisabeta Blvd, 030018, Sect. 3-Bucharest, Romania
Florica Ionescu
Research Development National Institute for Textile and Leather-Division Leather and Footwear Research Institute, 93 Ion Minulescu St., Sect 3, 031215-Bucharest, Romania
Simona Litescu
Department of Analytical Chemistry, Faculty of Chemistry, University of Bucharest, 4-12 Regina Elisabeta Blvd, 030018, Sect. 3-Bucharest, Romania
Ioana Vasilescu
Department of Analytical Chemistry, Faculty of Chemistry, University Politehnica of Bucharest, 1 Gheorghe Polizu St., Sect. 1, Cod 011061-Bucharest, Romania
Gabriel Lucian Radu
3Department of Analytical Chemistry, Faculty of Chemistry, University Politehnica of Bucharest, 1 Gheorghe Polizu St., Sect. 1, Cod 011061-Bucharest, Romania