Research Article
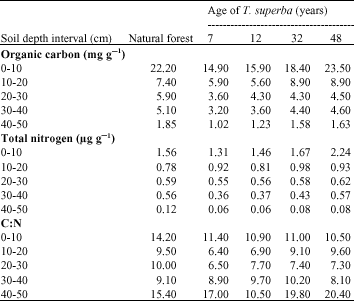
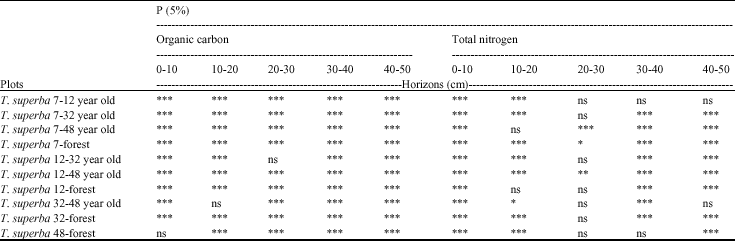
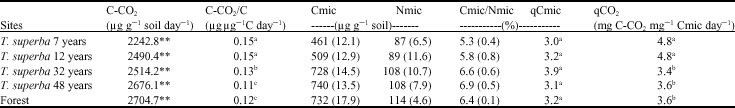
Soil Organic Matter and Biological Changes in a Natural-to Planted Forest Succession: Terminalia superba Plantations Grown on Deforested Plots in Congo
Institut de Developpement Rural, Universite Marien Ngouabi, BP 13502 Brazzaville, Congo
S. Makosso
Institut de Developpement Rural, Universite Marien Ngouabi, BP 13502 Brazzaville, Congo