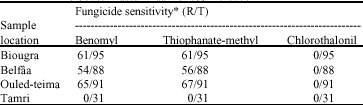
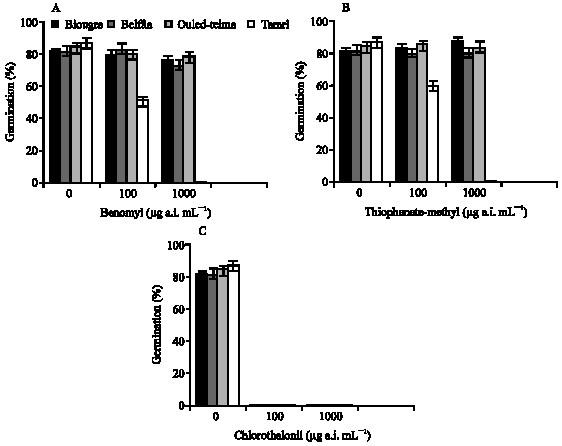
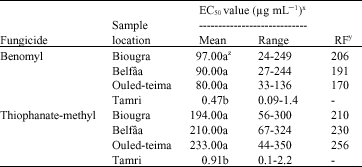
Research Article
Resistance of Verticillium theobromae to Benzimidazole Fungicides in Morocco
Laboratoire de Biotechnologie et de Valorisation des Ressources Naturelles, Faculte des sciences, B.P. 8106, Universite Ibn Zohr, Agadir, Maroc
B. Saadi
Laboratoire de Biotechnologie et de Valorisation des Ressources Naturelles, Faculte des sciences, B.P. 8106, Universite Ibn Zohr, Agadir, Maroc
E.H. Boudyach
Laboratoire de Biotechnologie et de Valorisation des Ressources Naturelles, Faculte des sciences, B.P. 8106, Universite Ibn Zohr, Agadir, Maroc
A. Ait Benaoumar
Laboratoire de Biotechnologie et de Valorisation des Ressources Naturelles, Faculte des sciences, B.P. 8106, Universite Ibn Zohr, Agadir, Maroc