Research Article
Relative Selectivity Coefficient as a New Concept for Evaluating Electrochemical Dopamine Sensors
Department of Chemistry, Faculty of Science, Zagazig University, Zagazig 44519, Egypt
Dopamine has the chemical formula (C8H11NO2). Its chemical name is 4-(2-aminoethyl)benzene-1,2-diol and it is abbreviated as DA (Benes, 2001). It belongs to the catecholamine family; it is a precursor to epinephrine (adrenaline) and then norepinephrine (noradrenaline) in the biosynthetic pathways for these neurotransmitters (Fig. 1).
Dopamine is a phenethylamine naturally produced by the human body. In the brain, dopamine functions as a neurotransmitter. Dopamine is also a neurohormone released by the hypothalamus. Its main function as a hormone is to inhibit the release of prolactin from the anterior lobe of the pituitary. It is commonly associated with the pleasure system of the brain, providing feelings of enjoyment and reinforcement to motivate a person proactively to perform certain activities.
 | |
| Fig. 1: | Structural formulas of dopamine (DA), N,N-bis-ethoxycarbonyl-1,10-diaza-4,7,13,16-tetraoxacyclo-octadecane (DZCE) and potassium tetra-kis-[3,5-bis-(trifloro-methyl)-phenyl] borate (K-TFPB) |
Deficiency in biosynthesis of dopamine will result in Parkinson`s disease. In contrast, the production of more dopamine is recorded for schizophrenic patients. Therefore, it is important to follow up the dopamine level in both cases during, before and after medications. Dopamine can be supplied as a medication that acts on the sympathetic nervous system, producing effects such as increased heart rate and blood pressure.
Different methods were used for the determination of dopamine. Flow Injection Analysis (FIA) and HPLC with chemiluminescence detection were applied for determination of DA by Nalewajko et al. (2007). Catecholamines were determined by the spectrophotometric method based on the use of poly (dimethylsiloxane) (PDMS) technology (Maminski et al., 2005). Abdulrahman et al. (2005) reported a flow injection-spectrophotometric determination of some catecholamine drugs via oxidative coupling reaction with p-toluidine and sodium periodate. Nour El-Dien et al. (2005) described two spectrophotometric methods for the determination of dopamine derivatives.
Glassy carbon electrodes were applied for the determination of dopamine. A sensitive and selective electrochemical method for the determination of dopamine (DA) was developed using a calix-4-arene crown-4 ether film modified glassy carbon electrode (Lai et al., 2007). The Si-TiPH bulk modified carbon paste electrode (Kooshki and Shams, 2007) and Nafion coated carbon paste electrode (Alpat et al., 2005) was used for the selective determination of dopamine (DA) in the presence of ascorbic acid. Electrocatalytic oxidation and selective detection of dopamine at a 5,5-ditetradecyl-2-(2-trimethyl-ammonioethyl)-1,3-dioxane bromide self-assembled bilayer membrane modified glassy carbon electrode was introduced by Lin and Gong (2004). Hayashi et al. (2003) succeeded in detecting dopamine (DA) in the presence of L-ascorbic acid using interdigitated electrode array. Cheng et al. (2005) applied fiberion carbon-fiber electrode for in vivo determination of concentration of cationic neurotransmitter dopamine.
Many researchers constructed amperometric sensors for dopamine. Cu-dipyridyl complex was applied for preparing amperometric sensors for dopamine (Sotomayor et al., 2002). Tu and Chen (2002) applied nano-disposable biosensors for dopamine. Plant tissue containing oxidizing enzyme was applied to build up biosensor for determination of dopamine and other catechol amines (Mazzei et al., 1992).
Some previous researches were reported for the determination of DA using ion-selective electrodes. Calix-3-arene derivative was applied for preparing DA-selective electrode (Saijo et al., 2007). Júnior et al. (2000) constructed flow injection analysis (FIA)-potentiometric sensor for dopamine based on poly (ethylene-co-vinyl acetate)-Cu (II) ion. Othman et al. (2004) prepared PVC-membrane electrodes based on 12-crown-4-phosphotungestic and 12-crown-4-TPB ionophores for dopamine determination. Dopamine was analysed either by an indirect method using periodate selective electrode (Montenegro and Sales, 2000) or by direct method using dopamine selective electrode (Lima and Montenegro, 1999) based on tetra-chlorophenylborate. Earlier, Liu and Yu (1990) applied crown ethers as neutral ionophores for preparing electrodes for a primary amines.
In fruits, dopamine is the substrate for polyphenol oxidases (PPOs). These are a family of enzymes responsible for browning of fresh fruits and vegetables when they are cut or bruised. This helps to protect damaged fruits and vegetables against growth of bacteria and fungi (Romphophak et al., 2005).
In the present study, two highly selective sensors were introduced for the determination of dopamine. A comparison between the properties of the diaza crown and the previously reported crown based electrodes was established. The new electrode allows the direct determination of dopamine without pre-separation using toxic solvents or expensive instrumentation like in HPLC. No need of liquid nitrogen, like in polarographic methods. Ascorbic and uric acids were not interfering in dopamine determination by this electrode which is recorded when using carbon paste electrodes. An improvement of selectivity was found after adding synthesized diaza-18-crown-6 ether (DZCE) derivative to the potassium tetrakis-[3,5-bis-(trifloromethyl) phenyl] borate (K-TFPB). Evaluation of selectivity properties was applied by introducing and discussing the idea of Relative Selectivity Coefficient “RSC” for the first time.
Synthesis of the host molecule: DZCE was synthesized according to the procedure described previously (Hodgkinson et al., 1979). It depends on the addition of (5.84 g) of N,N-bis-ethoxycarboxycarbonyl-1,8-diamino-3,6-dioxaoctane in dry dimethyl sulfoxide (DMSO) (75 cm3) dropwisely to a stirred suspension of sodium hydride (1.12 g) in dry dimethyl sulfoxide (125 cm3). After 3 h, triethylene glycol bis-p-sulfonate (10 g) in dry DMSO was added to the formed solution and the mixture was sit for 3 days under nitrogen. Then, HCl (2 M) was added and the mixture was extracted in CHCl3. The organic extract was washed with water, dried in MgSO4 and evaporated. The residual oil was purified by chromatography on silica. The macrocycle DZCE was obtained as solid (m.p. 75-78°C). The structure of the obtained compound was verified by elemental analysis and IR (found C, 52.9; H, 7.1; N, 8.4%; confirm C18H34N2O8; vmax 1680 cm-1).
Reagents and materials: Materials used for the preparation of electrodes were tetrahydrofuran (THF) (Merck) (after its distillation), dodecyl phthalate (DDP) (Fluka), potassium tetra-kis-[3,5-bis-(trifloromethyl) phenyl] borate (K-TFPB) (Fluka), high molecular weight poly (vinylchloride) (PVC) (Fluka) and N,N-bis- ethoxycarbonyl-1,10-diaza-4,7,13,16-tetraoxacyclo- octadecane (diaza-18-crown-6) (DZCE) (synthesized as aforementioned) were used for the preparation of all membranes. Dopamine (4-(2-aminoethyl)benzene-1,2-diol) (Sigma), ephedrineHCl (Sigma), adrenaline (Sigma), caffeine (Sigma), pilocarpine hydrochloride (Sigma), atropine sulfate (Sigma); adrenaline (Sigma), ascorbic acid, urea, glycine, arginine and sodium glutamate (Aldrich) were used. Dopamine injection ampoules (40 mg/5 cm3) were purchased from the local drug stores. Nitrate salts of inorganic cations (Na+, K+, Li+, NH4+, Ca++, Mg++ and Ba++) were purchased from (Fluka). De-ionized water was operated through the whole work for the preparation of different solutions and for rinsing the electrodes.
Instruments: The cell-EMF values were measured using a bench top model Sension-4 (HACH, USA). The instrument was loaded to a computer system through RS-232 connection and HACH-software. The same instrument was applied for pH-measurements.
The spectrophotometric measurements were carried by a UV-VIS-spectrophotometer DR-4000 (HACH, USA), fitted with flow injection unit and loaded to a computer system through RS-232 connection and HACH-software.
Electrode system and potentiometric measurements: One milligram KTFPB or (1 mg KTFPB + 2 mg DZCE) were applied as sensor materials for membranes I and II, respectively. The mentioned ionophores were mixed with 60-67 mg DDP plasticizer and 30-31.5 mg PVC. The membrane was prepared according to a procedure described before (Zareh et al., 2001). The membrane discs were mounted on electrode Phillips bodies (type IS 561, Eindhoven, Netherlands) for electromotive force measurements. The electrode was filled with an aqueous inner filling solution (KCl 0.01 M and DAHCl (0.01 M) solution). It was doped 24 h in 10-2 M, before first use.
The potentiometric measurements were performed at room temperature (25°C) in a Galvanic cell, which can be represented as the following:
Ag-AgCl/Li-acetate/Sample//Ion-selective membrane// Inner filling solution/Ag-AgCl
The outer compartment of the reference electrode was filled with lithium acetate solution (0.1 M).
The potential was measured by immersing the electrodes with a reference Calomel into water (50 cm3). Different aliquots of DAHCl (10-2 and 10-1 M) were added to cover a concentration range of DAH+ (10-7-10-2 M). The potential values were recorded and plotted versus p[DAH+] using Microsoft Excel 2003. For studying the pH-effect on the electrode potential, the pH was changed using NaOH or HCl (0.1 M). The potential values were recorded at different pH for the electrodes in 10-4, 10-3 and 10-2 M DAH+ solutions.
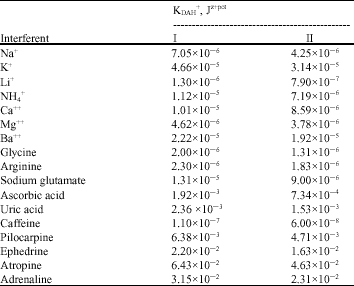
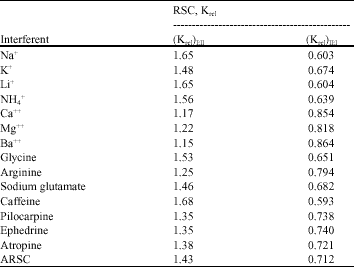
The selectivity coefficient values for several cations (Na+, K+, Li+, NH4+, Ca++, Mg++ and Ba++), aminoacids (glycine, arginine and sodium glutamate) and pharmaceutical amines (ephedrineHCl, adrenaline, caffeine, pilocarpine hydrochloride and atropine sulfate) are calculated (Table 1) by the use of the Separate Solution Method (SSM) (Guilbault et al., 1976). The emf of the interference solution (0.01 M) and that for the same concentration of DAH+ solution were measured. Then, the selectivity coefficient values of the electrodes (Ki,j pot) were estimated for the different interferents according to the equation:
log Kpot DAH+, Jz+ = (Ej — EDAH+) / S + [1-(ZDAH+/Zj)] log aDAH+ | (1) |
where, E represents the emf readings for the primary ion DAH+ and the interfering ion (Jz+) and (S) is the observed slope for the primary ion.
Injection ampoules of dopamine HCl (5 cm3) (products of EIPICO, Egypt; Ebewe Pharma, Austria; or Pierre Fabre, France) were diluted to 50 mL solutions. The obtained solutions were transferred to the potentiometic cell. Both DAH+-selective and the reference electrodes were immersed into the solutions and the cell EMF was measured. The potential readings of the sample solutions were compared to previously prepared calibration graph under the same condition.
Twenty gram (fresh weight) of sliced banana pulp (two days ripened) were transferred to a beaker containing 20 cm3 0.1 M HCl. The mixture was homogenized in a blender. The homogenate was centrifuged for 20 min. The supernatant solution was adjusted to pH 4 using K2CO3, then brought up to 50 cm3. The solution was transferred to the potentiometric cell and subjected for EMF-measurements using the proposed electrode. The obtained values were compared to a calibration graph of dopamine standard solutions treated typically like the measured samples.
For the spectrophotometric analysis, after extraction as aforementioned, purification step was applied. It was performed by elution of 5 cm3 of the extract into a Dowex 50 X-8 column, which was washed with 20 mL 2 M HCl, 5 mL water, 10 cm3 1 N acetate-acetic buffer (pH 6) and finally with 5 mL water. The dopamine was eluted by 6 mL 1 M HCl at rate of 0.25 cm3 min-1, followed by 6 mL 2 M HCl. The eluted dopamine was assayed according to procedures mentioned by Abdulrahman et al. (2005). It is based on the oxidative coupling with p-toluidine (0.008% w/v) and sodium periodate (0.4 mM) giving an orange dye with maximum absorption at 480 nm. The obtained results were compared to a calibration graph previously prepared for dopamine solutions (2-50 μg cm-3) under the same conditions.
Two electrodes with different membrane compositions were prepared. One has a membrane containing KTFPB (type-I) and the other constitutes (KTFPB+DZCE) (type-II). Both electrodes exhibit typical Nernstain slope (57.8 and 58.4 mV/decade). They work linearly down to 7.9x10-6 M DAH+. The calibration graphs representing both electrodes are displayed in Fig. 2. The lower linear limit of the calibration graph for electrode II is better than that of electrode I. This is because the ion-pair in electrode-II contains a cavity, which helps the chelation of DAH+. The DZCE works like the charge carrier that helps the ion-association with [TFPB]–.
The mechanistic equation that represents the exchange reaction at the membrane-solution interface for electrode-I is written as below:
[DAH]+ + [TFPB]– ===== [ TFPB– DAH+] |
| Keq = [ TFPB– DAH+] / [DAH+] [TFPB–] |
In case of electrode-II, the following equilibrium is expected:
[TFPB]– K+ + DZCE ===== [(DZCE-K)+ TFPB–] |
[(DZCE-K)+ TFPB–] + DAH+ Cl– ===== [(DZCE-DAH)+ TFPB–] + KCl |
Keq = [(DZCE-DAH)+ TFPB–] / ([(DZCE-K)+ TFPB–] [DAH+]) |
The dynamic response of both electrodes showed instantaneous and stable potential readings. Figure 3 displays the obtained results.
The mV-pH curves for both electrodes (I and II) are shown in Fig. 4. The working pH-range is 3.5-8.3 for both electrodes for 10-2 M DAH+.
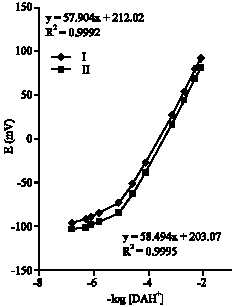 | |
| Fig. 2: | Calibration graphs for dopamine electrodes based on diazacrown ether derivative |
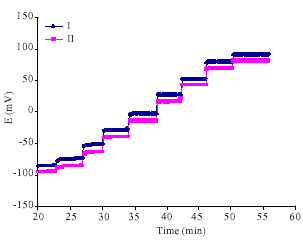 | |
| Fig. 3: | Dynamic response of dopamine electrodes based on diazacrown ether derivative |
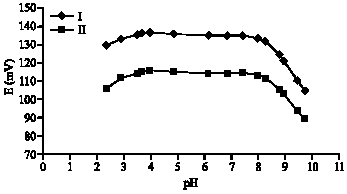 | |
| Fig. 4: | Effect of pH on the potential of dopamine electrodes based on either TFPB and (TFPB + DZCE) in 0.01 M dopamine solution |
| Table 1: | Selectivity coefficient values (KDAH+, Jz+pot) for both dopamine electrodes based on TFPB and diazacrown ether analogues |
 | |
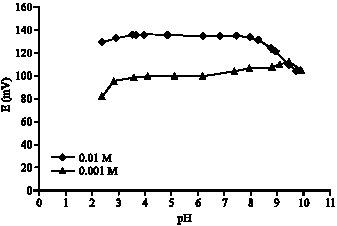 | |
| Fig. 5: | Effect of pH on the potential of dopamine electrode type-II based on K-TFPB and DDP into 0.01 and 0.001 M dopamine solutions |
Whenever 10-3 M DAH+ solution was measured, the plateau potential values were between 2.9 and 9.1. This is a wide pH-range compared to the previous electrodes, which are not working in alkaline medium (2.5-6 and 3.5-6). Figure 5 shows the mV-pH curves for the dopamine electrode-II at 10-3 and 10-2 M concentrations. The formation of the free base is the reason of the break in the basic part of the plateau. This depends on the concentration of DAH+. In acidic medium, the hydrogen ion interference is the reason of the curve break.
Selectivity and Relative Selectivity Coefficient (RSC): The selectivity coefficient values (KDAH+jz+ pot) for the common inorganic cations, amino acids, pharmaceutical amines were calculated and recorded in Table 1. It is shown that the values of the selectivity coefficients for the inorganic cations range between 10-5 and 10-7. This shows a super selectivity properties. Amino acids like glycine, arginine, glutamate showed values of the selectivity coefficient of the order of 10-5-10-6. This is because the presence of the Zwitter ion masked the amine group from being part of chelation with the DZCE. This favors the discrimination of DPH+ over them. In case of ascorbic acid, uric acid and other pharmaceutical amines (pilocarpine, adrenaline, ephedrine and atropine), the selectivity coefficient values were of the order of 10-2-10-3. Although these values are higher than those for other cations, they are still suitable for measuring dopamine in presence of them.
The relative selectivity coefficient Krel (RSC) is a parameter that is introduced to evaluate the selectivity properties of electrodes or sensors of different composition, but responding to the same ion. This factor helps the mathematical comparison between these sensors or electrodes. It can be calculated by using their selectivity coefficient values (Ki,j pot). For example if I and II are two electrodes responding to the same primary ion, the following equation can be applied to calculate RSC for both electrodes towards each other, for one interferent:
| (Krel)I:II = (Ki,j pot)I /(Ki,jpot)II | (2) |
| (Krel)II:I = (Ki,jpot)II / (Ki,jpot)I | (3) |
If there are (n) interferents, the Average Relative Selectivity Coefficient (ARSC) (K I:II)av, can be calculate as below:
| (Krel-I:II)av = [Σ1→n (Krel)I:II-n]/n | (4) |
| (Krel-II:I)av = [Σ1→n (Krel)II:I-n]/n | (5) |
As a general formula if there are (m) number of electrodes to be compared with (X) electrode under study, a general formula (Zareh`s equation) can be written:
| (Krel-X)total = [(Krel-X:1)av + (Krel-X:2)av + (Krel-X:3)av + (Krel-X:4)av + .....+ (Krel-X:m)av]/(m) | (6) |
where, (1, 2, 3, 4,....m): refers to the number of electrodes to be compared with; (Krel-X)total : is the average of the relative selectivity coefficient (TARSC) for X-electrode toward all electrodes. It can be predicted that the smaller the (Krel-X)total-value, the better the selectivity properties of an electrode.
The selectivity coefficient values are shown in Table 1. The values (KDAH+, Jz+) for electrode II is lower than those for electrode I. This means a better selectivity for electrode II than electrode I is expected. Nevertheless, this is not enough to discriminate between the selectivity of the two electrodes. Therefore, evaluation of selectivity properties was conducted by applying the aforementioned relative selectivity rules. Generally, it can be reported that the selectivity coefficient values of electrode II has numerical values less than that for electrode I by a factor 0.712. This factor is the value of the relative selectivity coefficient RSC for electrode II toward electrode I (Krel)II:I. For the inorganic cations, the average (Krel)II:I is 0.723, while for organic amines it is 0.703. Table 2, shows the obtained results. This shows that the selectivity properties of electrode II is better than that of electrode I toward these cations.
| Table 2: | Relative Selectivity Coefficient (RSC) values for both I and II dopamine electrodes towards each other |
 | |
This is caused —y the association of the formed ion-pair by the host DZCE-molecule, which led to more stability of the formed DAH+-complex.
Likewise, by using the RSC-concept the selectivity properties of electrode I can be compared to the previously reported electrodes (pre1) and (pre2) (Othman et al., 2004). Where, (pre1) and (pre2) refer to the previously reported electrodes with crown ether-phosphotungestic acid (CE-PTA) and crown ether-tetraphenyl-borate (CE-TPB) ion pairs, respectively. The total average relative selectivity coefficients (TARSC) values for electrode I are (Krel)I:pre1: 9.9x10-3 and (Krel)I:pre2: 5.0x10-3. By the same way electrode II is compared to previously reported electrodes (pre1) or (pre2). It is found that (Krel)II:pre1 is 6.7x10-3 and (Krel)II:pre2 is 3.4x10-3. This means that the present electrode exhibits better selectivity properties than the previously reported electrodes. Table 3 shows the calculated values.
The application of the proposed electrode for the determination of dopamine in its pharmaceutical preparations was established. Samples of injection solutions from different companies were subjected to the electrode analytical procedures. Table 4 shows the obtained results. The recovery showed values between 98.5 and 99.7%. The RSD values ranged between 1.32 and 1.64. The obtained results agreed with those obtained by applying the previously reported spectrophotometric method (Abdulrahman et al., 2005).
The electrode was applied for determination of dopamine in banana. The results agreed with those obtained by the mentioned spectrophotometric method. Table 5 summarizes the obtained results for dopamine in banana pulp after 2 days ripening.
| Table 3: | Relative Selectivity Coefficient (RSC) values for the present electrodes I and II toward the previously reported (Othman et al., 2004) electrodes (pre1) or (pre2) based on either (CE-PTA) or (CE-TPB), respectively |
 | |
| *Othman et al. (2004) | |
| Table 4: | Determination of dopamine in pharmaceutical samples using the proposed electrode type-II |
 | |
| *Abdulrhman et al. (2005), **Four determinations | |
| Table 5: | Determination of dopamine in banana pulp samples after two days ripening using the proposed electrode type-II |
 | |
| *Abdulrhman et al. (2005), **Relative standard deviation (4 determinations) | |
Application of originally formulated diazacrown ether derivative as a new ionophore for preparing dopamine electrode showed successful results. The selectivity and the sensitivity of electrodes based on the synthesized diazacrown ethers were better than those for previously reported electrodes based on the usual crown ethers.
The use of the new concept of the relative selectivity coefficient is very useful in judgment between the different electrodes that are selective to the same ion. It made the comparison more accurate and specific.