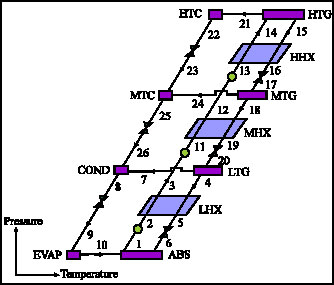
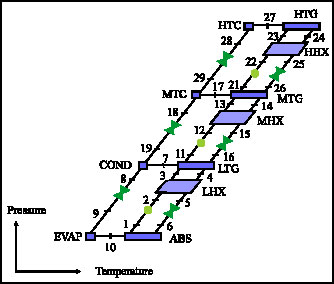
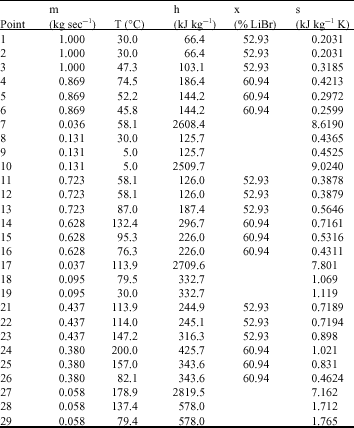
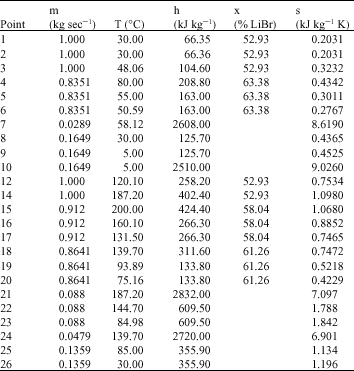
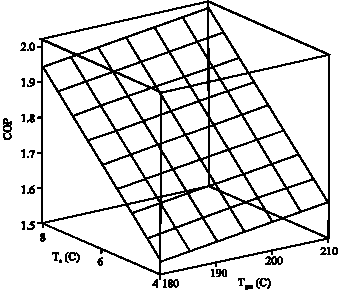
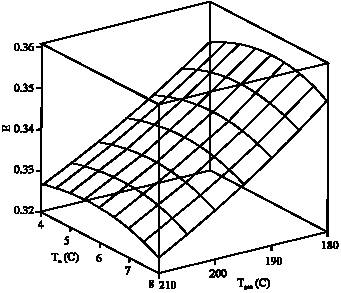
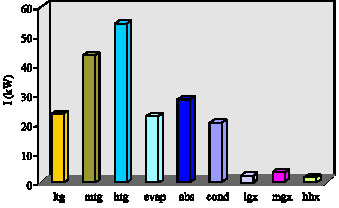
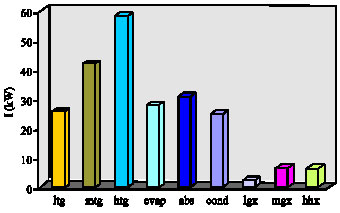
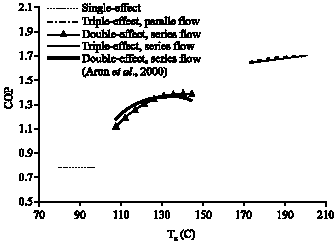
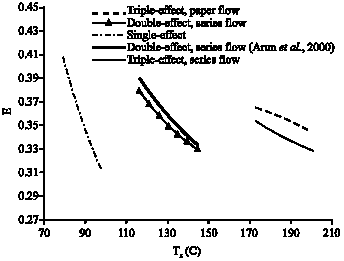
Research Article
Performance Comparison of Triple-Effect Parallel Flow and Series Flow Absorption Refrigeration Systems
Faculty of Mechanical Engineering, Tabriz University, Tabriz, Iran
F. Ranjbar
Faculty of Mechanical Engineering, Tabriz University, Tabriz, Iran
S.M. Seyed Mahmoudi
Faculty of Mechanical Engineering, Tabriz University, Tabriz, Iran