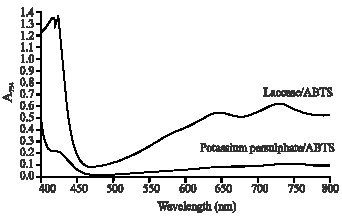
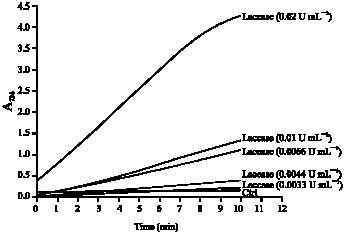
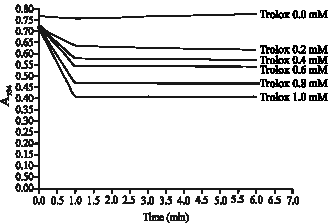
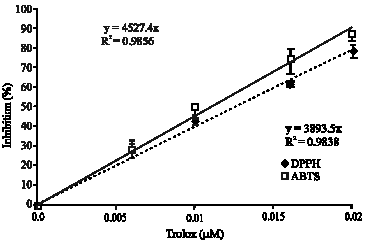
Research Article
Laccase-Aided Antioxidant Activity Assay and Antioxidant Activity of Selected Thai Vegetables
Protein and Enzyme Technology Research Unit Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham 44150, Thailand
R. Sarnthima
Protein and Enzyme Technology Research Unit Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham 44150, Thailand