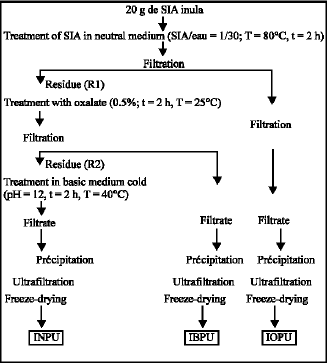
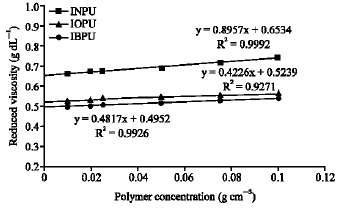
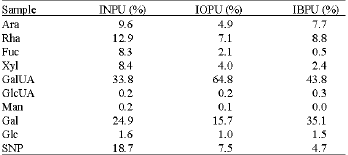
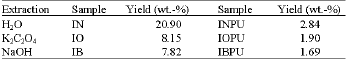Research Article
Fraction of Soluble Polysaccharides from Inula crithmoides by Sequential Extraction
Laboratoire Polymères, Biopolymères et Matériaux Organiques, Faculté des Sciences de Monastir, 5000 Monastir, Tunisia
Hatem Majdoub
Laboratoire Polymères, Biopolymères et Matériaux Organiques, Faculté des Sciences de Monastir, 5000 Monastir, Tunisia
Sadok Roudesli
Laboratoire Polymères, Biopolymères et Matériaux Organiques, Faculté des Sciences de Monastir, 5000 Monastir, Tunisia