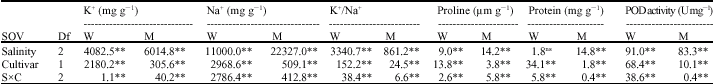
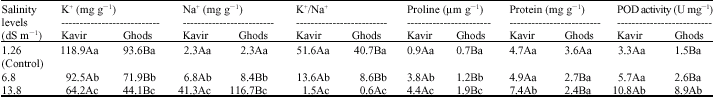
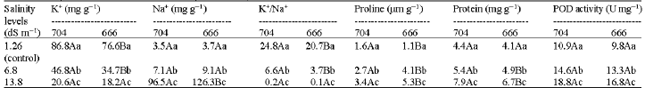
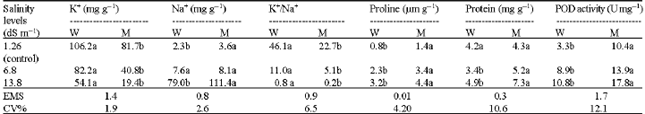
Research Article
Comparison Between Salt Tolerance of Various Cultivars of Wheat and Maize
Department of Crop Production and Plant Breeding, College of Agriculture, Shiraz University, Shiraz, Iran
H. Pakniyat
Department of Crop Production and Plant Breeding, College of Agriculture, Shiraz University, Shiraz, Iran