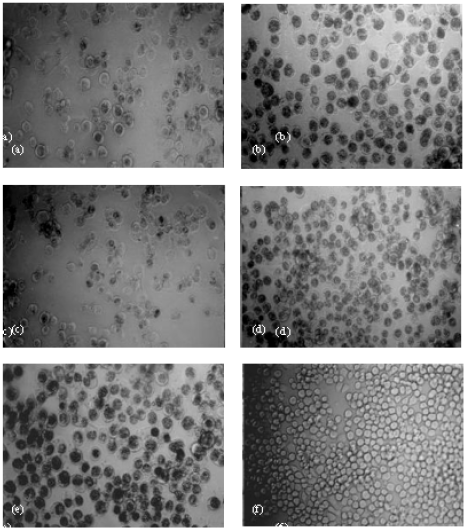
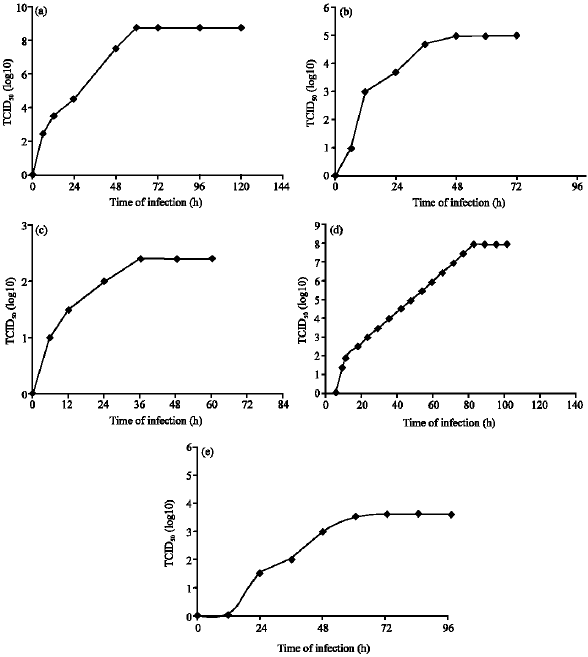
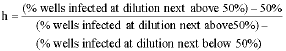Research Article
Replication of Syngrapha falcifera Multiple-Nuclear Polyhedrosis Virus-D in Different Insect Cells
Key Laboratory of Pesticide and Chemical Biology, Institute of Entomology,Huazhong Normal University, Ministry of Education, Wuhan 430079, China
Peng Jian Xin
Key Laboratory of Pesticide and Chemical Biology, Institute of Entomology,Huazhong Normal University, Ministry of Education, Wuhan 430079, China