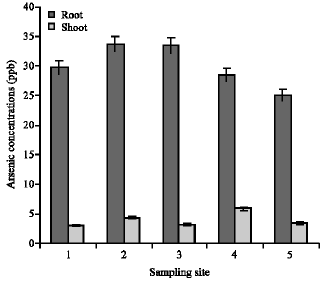
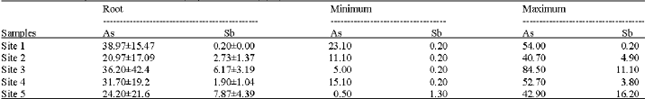
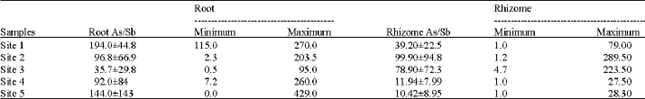
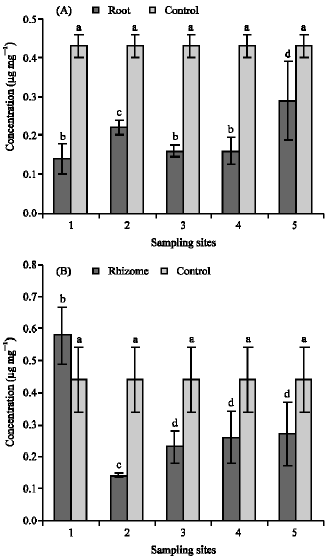
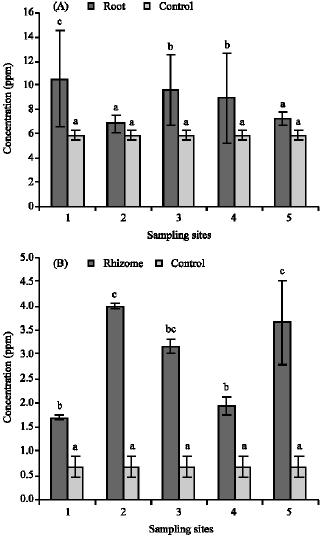
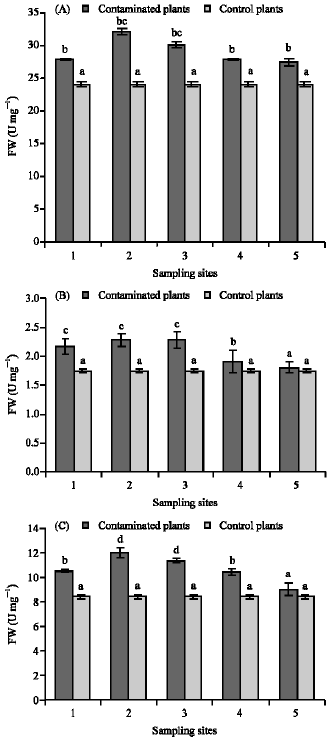
Research Article
Removing Arsenic and Antimony by Phragmites australis: Rhizofiltration Technology
Department of Biology, Faculty of Sciences, Water and Environmental Center,Ferdowsi University of Mashhad, Iran
H. Yousefzadeh
Department of Biology, Faculty of Sciences, Payam-Noor University, Tehran, Iran
M.H. Arbab-Zavar
Department of Chemistry and Environmental Chemistry Center, Ferdowsi University of Mashhad, Iran