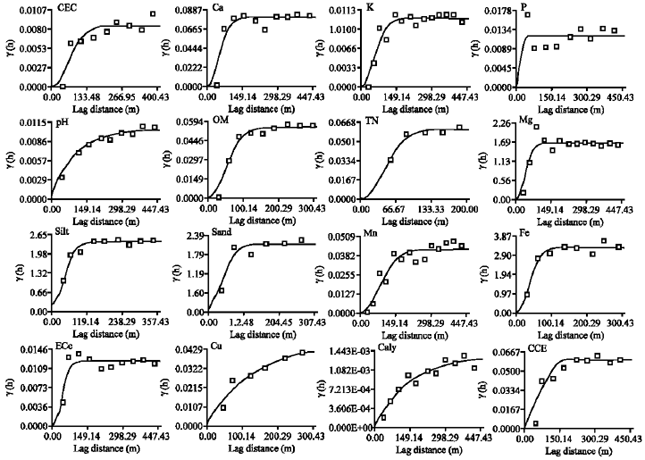
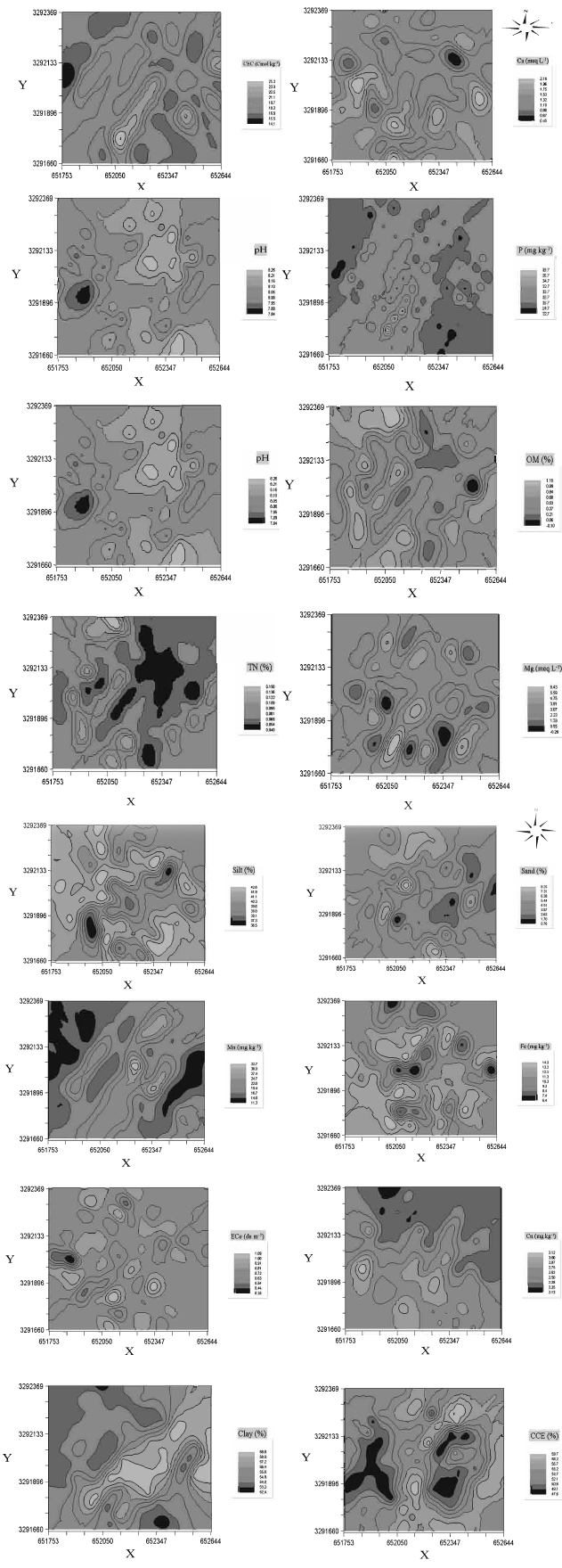
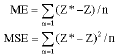Research Article
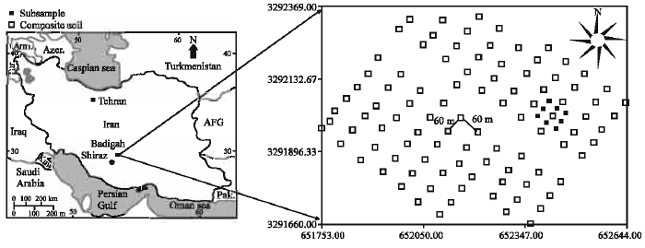
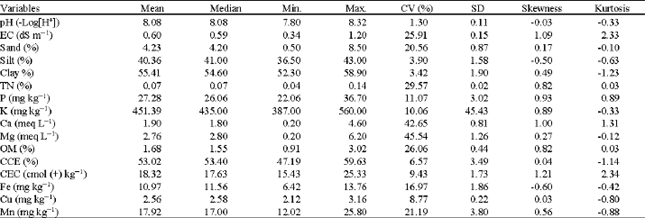
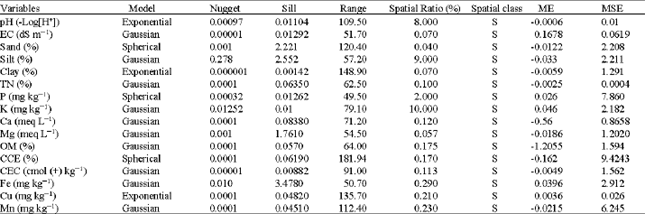
Spatial Variability of Soil Fertility Properties for Precision Agriculture in Southern Iran
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran
Mahboub Saffari
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran
Hamed Fathi
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran
Najafali Karimian
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran
Mostafa Emadi
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran
Majid Baghernejad
Department of Soil Science, College of Agriculture, Shiraz University, Shiraz, Iran