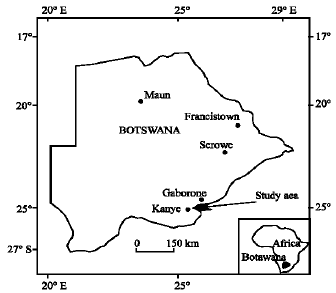
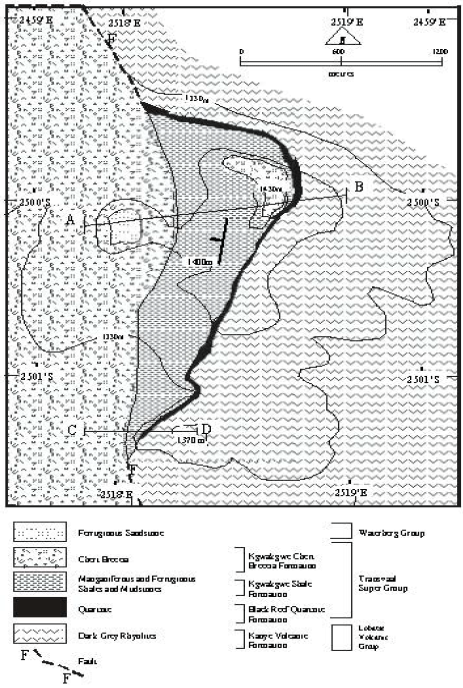
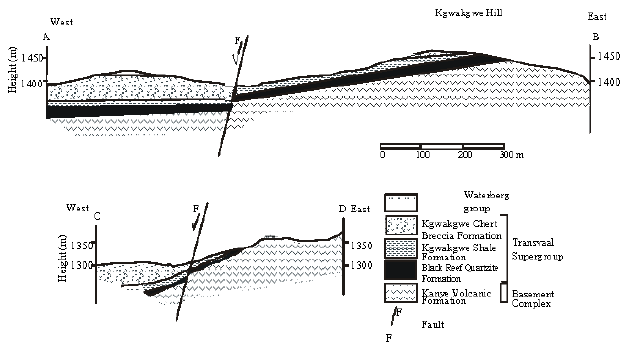
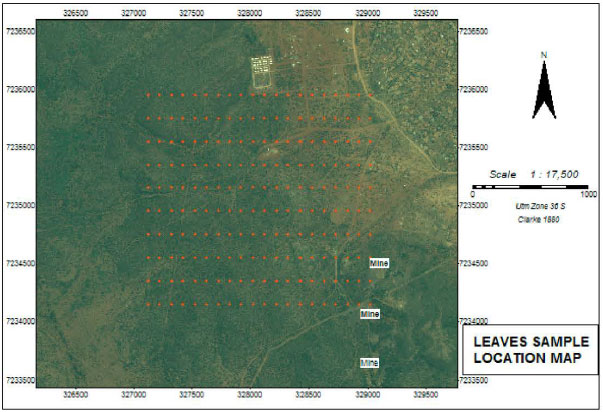
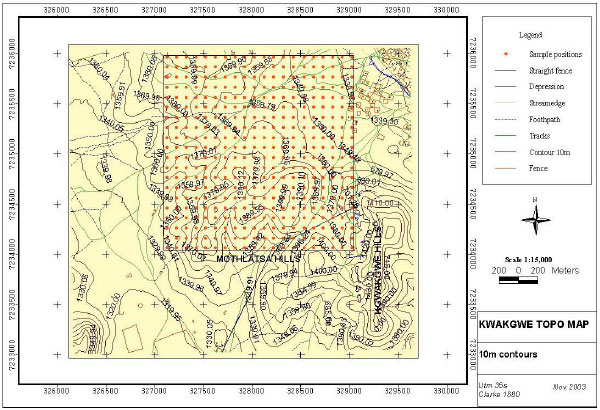
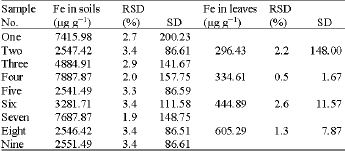
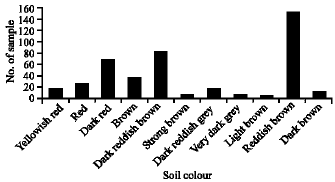
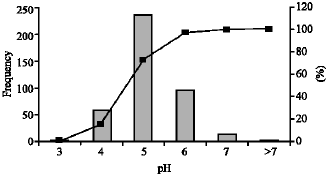
Research Article
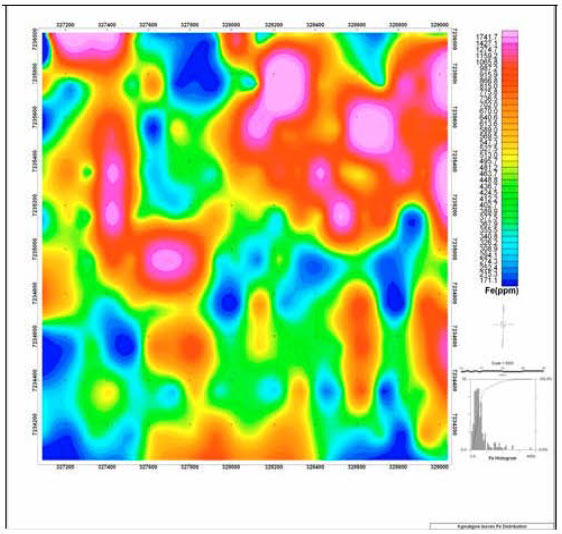
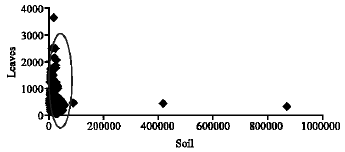
Spatial Distribution of Iron in Soils and Vegetation Cover Close to an Abandoned Manganese Oxide Ore Mine, Botswana
Department of Geology, Mining and Minerals Programs, University of Limpopo, Sovenga, P/B X1106 Limpopo Province, 0727, South Africa