Research Article
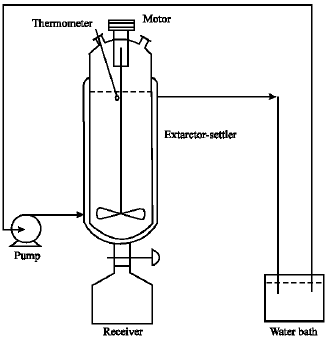
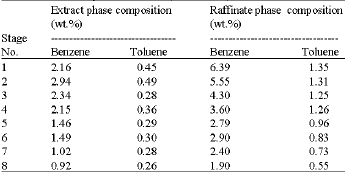
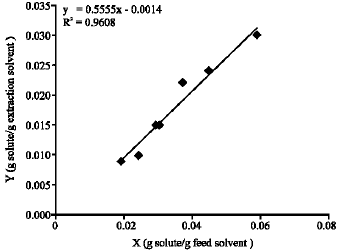
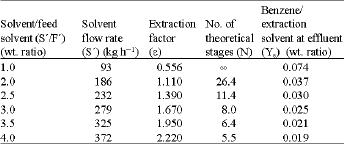
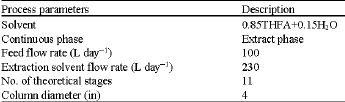
Liquid Extraction of Aromatic Hydrocarbons by Tetrahydrofurfuryl Alcohol, An Environmentally Friendly Solvent
lnstitute of Chemical Technologies, Iranian Research Organization for Science and Technology, P.O. Box 15815-3538, Tehran, Iran
D. Sadeghi Fateh
lnstitute of Chemical Technologies, Iranian Research Organization for Science and Technology, P.O. Box 15815-3538, Tehran, Iran
H. Shokouhi
lnstitute of Chemical Technologies, Iranian Research Organization for Science and Technology, P.O. Box 15815-3538, Tehran, Iran
A. Shahvelayati
Faculty of Science, Shahr-e Rey Branch, Islamic Azad University, P.O. Box 18735-334, Tehran, Iran
F. Golmohammad
lnstitute of Chemical Technologies, Iranian Research Organization for Science and Technology, P.O. Box 15815-3538, Tehran, Iran










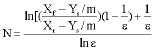
Fadoua FARGHI Reply
The tiltle of this article is interesting