ABSTRACT
Ruthenium complexes containing ring-substituted bipryidine ligands and related structures have longbeen known to efficiently catalyze water oxidation by strong chemical oxidants and at electrode surfaces. However, despite considerable effort, the goal of identifying the underlying molecular mechanisms has not yet been realized. In this paper, ZINDO/1 theory has been used to study a Ru complex found to react with water and Ce(NH 4)2(NO3)6 to liberate molecular oxygen (O2). The complex indicates a robust catalyst as compared to other earlier reported systems also a proposal mechanism for water oxidation is reported.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2007.451.454
URL: https://scialert.net/abstract/?doi=jas.2007.451.454
INTRODUCTION
Water oxidation to evolve O2 is an important and fundamental chemical reaction in photosynthesis. This reaction is catalyzed by a unique manganese enzyme referred to as Oxygen Evolving Complex (OEC), whose active site is comprised of an oxobridged tetramanganese cluster (Loll et al., 2005).
In past few years, there has been a tremendous surge in research on the synthesis of various manganese complexes (Limburg et al., 1999) aimed at simulating (OEC) of PSII.
Ruthenium-based catalysts also show promise for water oxidation. One such system is the blue dimer cis, cis-[{Ru-(bpy)2(H2O)}2O](ClO4)4 (bpy) 2,2’-bipyridine) in which the two Ru fragments are linked by a μ-oxo bridge which is prone to cleavage, thus limiting the lifetime of the catalyst (Hurst, 2005). This problem has been addressed by the design of ligands that bridge the two metal centers and thus increase the stability of the catalyst.
Recently, Zong and Thummel (2005) have synthesised new family of ruthenium complexes for water oxidation. In this paper a similar ruthenium complex is synthesised. This complex and Ce(NH 4)2(NO3)6 are found to react with water to liberate molecular oxygen (O2). The complex indicates a robust catalyst as compared to other previously reported systems
MATERIALS AND METHODS
Experimental Section (Eq. 1) (Zong and Thummel, 2005): trans-[Ru(4,4’ -bpy)2(OH2)](ClO4)2 (2). A suspension of 1 (167 mg, 0.5 mmol) in absolute ethanol (20 mL) and an ethanolic solution (10 mL) of RuCl3-3H2O (150 mg, 0.58 mmol) were mixed at room temperature and the mixture was refluxed for 1.5 h. To the mixture were added 4,4’ -bpy (78 mg, 0.5 mmol), water (10 mL) and triethylamine (0.2 mL) and the mixture was refluxed for 43 h.
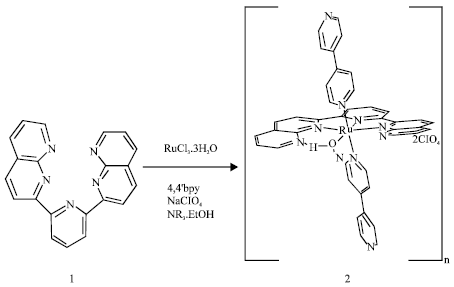 | (1) |
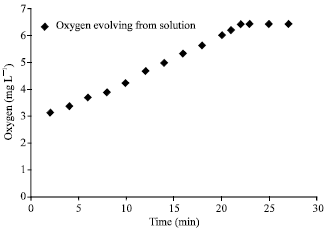 | |
| Fig. 1: | Time- dependence of O2 -evolution (0.1 g) (NH4)2 [Ce(NO3)6] was dissolved in 10 mL water after the deaeration (with nitrogen). 0.1 mg complex as solid suspension was added and O2 evolution was recorded with a oxygen meter under stirring (T = 35.6°) |
The solution was concentrated to about 10 mL, followed by the addition of aqueous NaClO4. The precipitate was filtered and washed with water, ether and acetone. IR (KBr, cm-1) 3444, 1602, 1415, 1085 and 804. Elemental analyzer is consistent with 2.
Water oxidation experiment was carried out with the complex and (NH4)2 [Ce(NO3)6] as oxidizing agent. When the complex or (NH4)2 [Ce(NO3)6] was dissolved in water, no evolution of gas was observed. 0.1 g (0.15 mmol) (NH4)2 [Ce(NO3)6] was dissolved in 10 mL water after the deaeration (with nitrogen). 0.1 mg complex as solid suspension was added and O2 evolution was recorded with an oxygen meter under stirring (Fig. 1). Total volume of oxygen is 0.65 mL (volumetric analysis). After oxygen evolution experiment catalyst didn’t show any change (IR) and can be used again after washing with HClO4.
Computational details: Calculations were made using ZINDO/1 method by personal computer.
RESULTS AND DISCUSSION
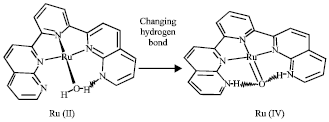
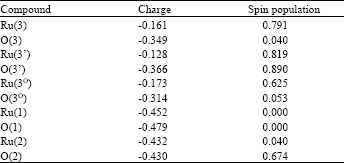
Proton is released in several states in the mechanism (Fig. 2). It can stabilize Ru(IV) complex. If a proton isn’t released (in high acidic concentration), Oxidation of the complex will increase charges of Ru and water molecule attached to Ru (Table 1). It can show important role of proton in regulating charge and stabilization of intermediates. Hydrogen bonds can be important in oxygen evolution (Najafpour, 2006a,b). Proposal mechanism for oxygen evolution in this complex is shown in Fig. 2. The Ru(III)-OH2 is a strong acid (pKa ~ 0.85) but Ru(III)-OH is a weak acid (pKa >9) also the ligand has a base group (pyridine ~pKa >9) and can be protonated (3,3’ in Fig. 3) hydrogen bonds between these protons and Ru(IV) = O (Changing hydrogen bond (Eq. 1)) can stabilize Ru(IV) = O that is important in oxygen evolution. Oxo group in this compound may have radical characterization (Xiaofan and Mu-Hyun, 2006).
 |
To test this theory for this complex, calculations were made using ZINDO/1 method. These results were shown in Fig. 3 and Table 1.
| Table 1: | Compared charges of Ru and oxo in 3’,3 and 3O. Calculations were made using ZINDO/1 method. Axial ligands are py (Fig. 3) |
 | |
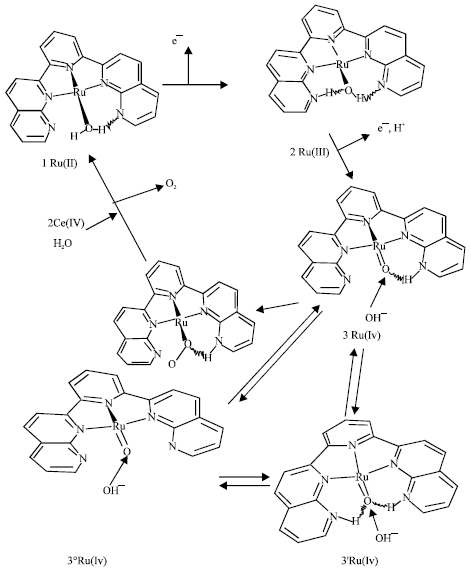 | |
| Fig. 2: | Proposal mechanism for oxygen evolution (Ce(IV) can oxidize hydrogen peroxide (Greenwood and Earshaw, 1984). Two pyridine (axial ligand) are deleted |
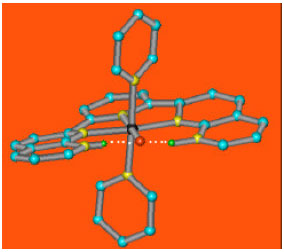 | |
| Fig. 3: | Structure for 3’(blue(C), yellow(N), red(O), green(H)black(Ru)(Calculations were made using ZINDO/1 method) |
3’ (Fig. 3) can be an important intermediate in oxygen evolution because of high spin population on oxo group (Ru = O). Rradical oxo can be suggested as an important intermediate in oxygen evolution (Xiaofan and Mu-Hyun, 2006).
CONCLUSIONS
A Ru complex and Ce(NH 4)2(NO3)6 are found to react with water to liberate molecular oxygen (O2). The complex indicates a robust catalyst as compared to other previously reported complexes. Also calculations were made using ZINDO/1 method. Calculations show important role of proton in regulating charge also this calculations Show an intermediate with high spin population on oxo group. This radical oxo can be important in oxygen evolution.
ACKNOWLEDGMENT
This study was supported by the Dorna Institute of Science.
REFERENCES
- Hurst, J.K., 2005. Water oxidation catalyzed by dimeric oxo bridged ruthenium diimine complexes. Coordinat. Chem. Rev., 249: 313-328.
Direct Link - Limburg, J., J.S. Vrettos, L.M. Liable-Sands, A.L. Rheingold, R.H. Crabtree and G.W. Brudvig, 1999. Functional model for O-O bond formation by the O2-evolving complex in photosystem II. Science, 303: 1524-1527.
Direct Link - Loll, B., J. Kern, W. Saenger, A. Zouni and J. Biesiadka, 2005. Towards complete cofactor arrangement in the 3.0 A resolution structure of photosystem II. Nature, 438: 1040-1044.
Direct Link - Najafpour, M.M., 2006. Role of Arg (357) in water oxidation in photosystem II. Pak. J. Biol. Sci., 9: 526-529.
CrossRefDirect Link - Najafpour, M.M., 2006. Current molecular mechanisms of photosynthetic oxygen evolution. Plant Biosyst., 140: 163-170.
Direct Link - Yang, X. and B. Mu-Hyun, 2006. cis,cis-[(bpy)2RuVO]2O4+ catalyzes water oxidation formally via in situ generation of radicaloid Ru(IV)-O!. J. Am. Chem. Soc., 128: 7476-7485.
Direct Link - Zong, R. and R.P. Thummel, 2005. A new family of Ru complexes for water oxidation. J. Am. Chem. Soc., 127: 12802-12803.
Direct Link








