Research Article
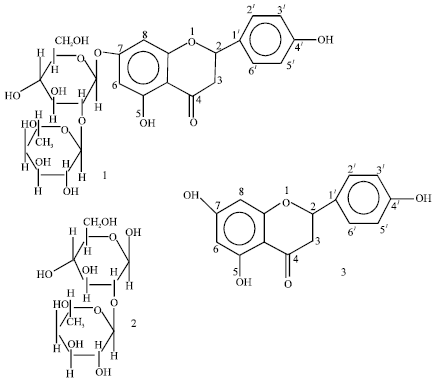
Isolation and Characterization of Flavanone Glycoside 4I,5, 7-Trihydroxy Flavanone Rhamnoglucose from Garcinia kola Seed
Department of Chemistry, Michael Okpara Univeristy of Agriculure Emudike, P.M.B 7267, Umuahia Abia state, Nigeria
F.N.I. Morah
Department of Chemistry, University of Calabar Cross River State, Nigeria