Research Article
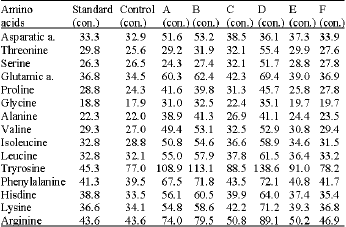
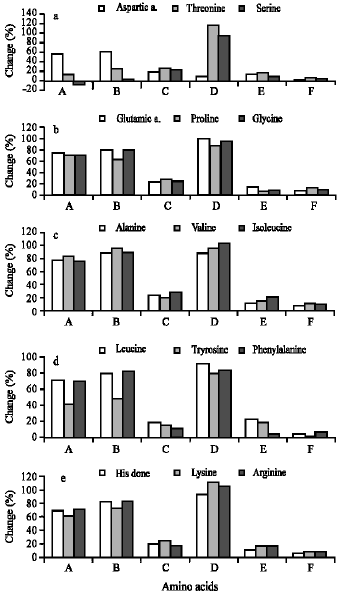
Induced Changes in the Amino Acid Profile of Biomphalaria alexandrina Molluscan Host to Schistosoma mansoni Using Sublethal Concentrations of Selected Plant Molluscicides
Department of Medicinal Chemistry, National Research Center, Dokki, Cairo, Egypt
Afaf El-Ansary
Department of Biochemistry, King Saud University, P.O. Box 22452, Riyadh,Zip Code 11495, Saudi Arabia