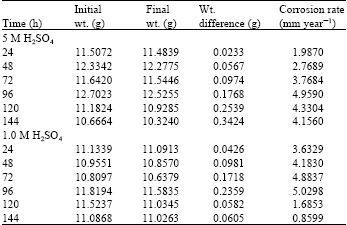
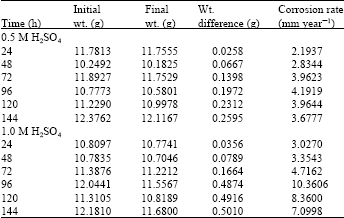
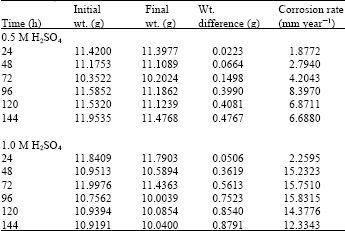
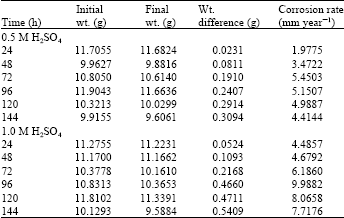
Research Article
The Effects of Zinc Additions on the Corrosion Susceptibility of Aluminium Alloys in Various Tetraoxosulphate (VI) Acid Environments
Department of Industrial Physics, Ebonyi State University, P.M B. 053, Abakaliki, Ebonyi, Nigeria
N.E. Idenyi
Department of Industrial Physics, Ebonyi State University, P.M B. 053, Abakaliki, Ebonyi, Nigeria
A.E. Umahi
Department of Industrial Physics, Ebonyi State University, P.M B. 053, Abakaliki, Ebonyi, Nigeria