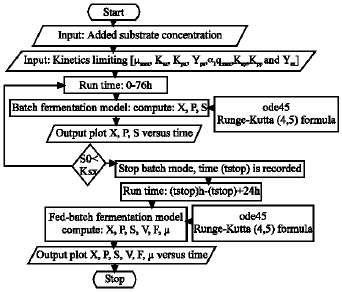
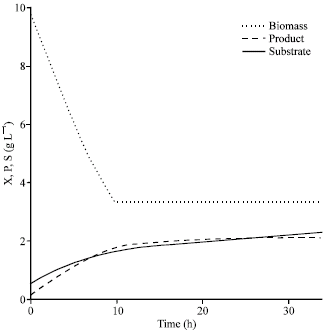
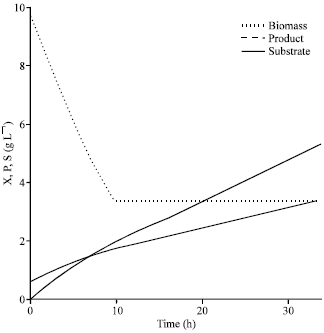
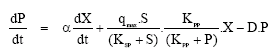Research Article
Fed-batch Fermentation of Lactic Acid Bacteria to Improve Biomass Production: A Theoretical Approach
Center of Materials and Minerals, School of Engineering and Information Technology, Universiti Malaysia Malaysia, Locked bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
Heng Jin Tham
Center of Materials and Minerals, School of Engineering and Information Technology, Universiti Malaysia Malaysia, Locked bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
Eng Seng Chan
Center of Materials and Minerals, School of Engineering and Information Technology, Universiti Malaysia Malaysia, Locked bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia