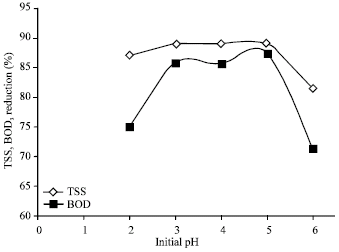
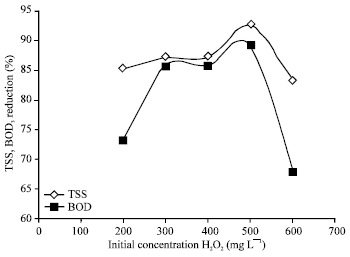
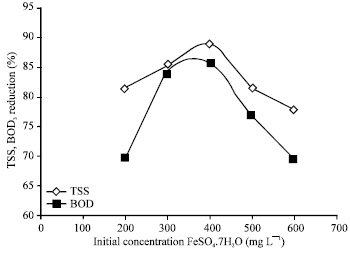
Research Article
Treatment of Pulp and Paper Mill Effluent Using Photo-fenton`s Process
Chemical Engineering Programme, School of Engineering and Information Technology, Universiti Malaysia Sabah,88999 Kota Kinabalu, Sabah, Malaysia
Melissa L. Gilbert
Chemical Engineering Programme, School of Engineering and Information Technology, Universiti Malaysia Sabah,88999 Kota Kinabalu, Sabah, Malaysia
Jidon Janaun
Chemical Engineering Programme, School of Engineering and Information Technology, Universiti Malaysia Sabah,88999 Kota Kinabalu, Sabah, Malaysia