Research Article
Characteristics of Lead Sorption by Zeolite Minerals
Department of Soil Science, College of Food Sciences and Agriculture, King Saud University, P.O. Box 2460, Riyadh 11451, Saudi Arabia
Soils and water resources were approved to be contaminated with heavy metals, as a result of increasing industrialization and mining activities (Al-Farraj and Al-Wabel, 2006). Increasing levels of heavy metals including Pb2+ in the environment, represent a serious threat to human health, living resources, soil quality and ecological systems (He, 1998; Kham et al., 2000; Nigam et al., 2001). Thus, a great deal of attention should be paid to overcome and/or avoiding environment against lead and other heavy metals pollution. Firstly, understanding the behavior of heavy metals, especially lead is important for minimizing the risk of soil and water contamination with such metal. In this respect, information regarding the mechanisms of lead adsorption/desorption in soil minerals is important as these reactions control the strength of metal-soil surface interactions. Moreover, the slow desorption of Pb2+ in soils has been a major impediment in estimating the environmental hazard and the successful phytoremediation of contaminated sites with such metal (Mench et al., 1994; Salt et al., 1995; Wu et al., 2003). Also, understanding the processes governing the migration and plant availability of trace metals in soils is essential for predicting the environmental impact of spreading metal-containing wastes on agriculture land. The adverse effects of heavy metals including Pb2+ are related to the soils ability to adsorb and release these elements. A fair number of investigations indicated that, clay minerals, due to their low permeability, could play an important role, as physical barriers, for the isolation of metal from metal rich wastes (La Grega et al., 1994). Also, they could be used as a chemical barrier too, as consequence of the ability of some types of clay minerals to adsorb heavy metals and to avoid their environmental dispersion (Vega et al., 1995). In this regard, naturally occurring zeolites hold great potential for use as packing material in subsurface reactive barriers intercepting ground water plumes and for fixed bed reactors designed to remove heavy metals from industrial wastewater (Bowman et al., 1995; Steimle, 1995). Such minerals group is one of the Tectosilicates that characterized by three dimensional framework structure with higher cation exchange capacity and a cage-like structure that offers large internal and external surface areas for ion exchange, (Ming and Mumpton, 1989; Bowman et al., 1995). In another study Gatima et al. (2006), pointed out that the adsorption of lead or lead removal from lead contaminated soils was depended upon the cation exchange capacities of the used red mud, bone meal and pulverized fly ash. As, the CEC could be a major parameter in the adsorption capacity of the soil. One of the most abundant natural zeolite minerals is clinoptilolite, has characteristic tabular morphology shows an open reticular structure of easy access, formed by open channels of 8±10 member rings (Mondale et al., 1995). Moreover, such mineral has been shown to selectively remove cesium and strontium ions from radioactive wastewater (Lukae and Foldesova, 1994) and its use for removing other dissolved heavy metal cations is receiving increasing attention in this respect, considerable research has been conducted to characterize the chemical, surface and ion exchange properties of clinoptilolite (Ackley and Yang, 1991; Carland and Aplan, 1995; Mondale et al., 1995; Semmens and Martin, 1988; Zamzow and Murphy, 1992; Trasevich et al., 1997; Alexander and Tsadilas, 2003; Yang et al., 2005; Sprynskyy et al., 2006). However, there is a shortage in the information concerning the characteristics of Lead sorption by the other zeolite minerals. Therefore, the objectives of this study were to investigate the kinetics of Pb2+ sorption by variable zeolite minerals. The possibility to use such zeolite minerals in remediation of lead polluted areas was evaluated.
Zeolite samples: Four zeolite minerals namely, clinoptilolite, analcime, philipsite and chabazite were taken from (South west USA, Barstow California, PineValley Nevada and Christmas, Arizona), respectively. In each sample cation exchange capacity was determined with the method for arid zone soils described by Rhoades (1982). Surface area was measured using Ethylene Glycol Mono Ethyl Ether (EGME) adsorption according to the method described by Carter et al. (1965). The chemical compositions of the tested Zeoilite samples were determined by the method described by Moore and Reynolds (1997) and Tarasevich et al. (1997) (Table 1).
Adsorption of Pb2+: An adsorption/desorption experiment was conducted-in March 20, 2006, under the growth chamber of the Department of Soil Science, College of Food Sciences and Agriculture, King Saud University, Riyadh, Saudi Arabia. Twenty milligram from each of the studying clayminerals were separately weighed into duplicate centrifuge tubes and each mixed with 40 mL solution of 0.0, 0.1, 0.5, 1.0, 2.0, 3.0 and 5 Pb μ mol mL–1 as Pb (NO3)2 in solution with an ionic strength of 10 μmol L–1 Ca (NO3)2. The isotherm data were obtained at pH which were not kept constant, but were verified to remain <6.5 (Straw et al., 1998; Shen et al., 1999; Mario et al., 2002). The tubes were flushed with N2 gas for 1 min and capped tightly before being shaken at 60 rpm at 23°C for four days. The pH values were measured thereafter, centrifugation (3000 rpm for 15 min, at 25°C). Then the supernatant was taken from each tube for the determination of Pb using the ICP-AES, (Prkin Elemer, Model 4300 DV). Lead sorbed by the clay minerals was calculated as the difference between the initial Pb2+ and equilibrium Pb2+ concentrations. The amount of native adsorbed Pb2+ initially present in the used minerals was estimated by extrapolating the adsorption isotherms derived from higher to zero concentration of soluble Pb2+ and from the amount of Pb2+ extracted with 10 μmol L–1 Calcium nitrate Ca (NO3)2. The native Pb2+ so estimated was taken into account to establish the corrected adsorption isotherm. The data of Pb2+ sorption were firstly calculated according to Langmuir equation. The linear form of the isotherm used was:
(c)/(x/m) = 1/b (c) +1/kb, |
| (c) | = | Equilibrium concentration (μmol Pb2+ mL–1), |
| (x/m) | = | Adsorbed Pb2+(μg Pb2+ g–1 soil), |
| b | = | Dsorption maximum (μg Pb2+ g–1 soil) and |
| k | = | A constant related to bonding energy (mL μg–1). The data were also described by the Freundlich equation. The linear form of the isotherm used was: |
log (x/m) = 1/n log (c) + log k d |
Where 1/n is the slope of the regression line and kd and n are empirical constants.
| Table 1: | The chemical and mineralogical properties for the used clay minerals |
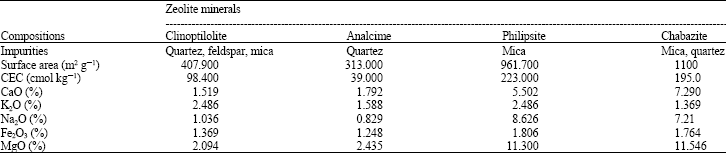 | |
Desorption of Pb2+: Desorption of Pb2+ was accomplished by repeated replacement with metal-free Ca (NO3)2 solution following adsorption. The tube with the mineral residue separated from the supernatant solution by the centrifugation was weighed to measure the residual Pb2+ in the mineral solution. Forty milliliter of 10 mM L–1 Ca (NO3)2 were added to each tube containing the Pb-enriched minerals residue. The suspensions were shaken at 200 rpm for 24 h at 25°C. The equilibrated suspensions were then centrifuged at 3000xg relative centrifugal force for 15.0 min. Thereafter, 20 mL of the supernatant were removed and replaced by 20 mL of 10 mM L–1 Ca(NO3)2. The procedure was repeated four times, resulting in a total four desorption for each adsorption sample tested. The non-extractable fraction of the adsorbed Pb2+ was obtained by the difference between the total recovered Pb2+ by four successive extractions with Ca (NO3)2 solutions.
Quality control: All the used chemicals were purchased from E. Merck (A.R., 99.9%). The apparatus, tubes and vessels were previously washed with 30% HNO3, then rinsed three times with deionized water and dried in an oven. All stages of sample preparation, analysis and measuring were carried out in a clean environment.
Properties of the used zeolite minerals: Data presented in Table 1 show the chemical and mineralogical properties of the used zeolite minerals. Apparently, the used minerals are varied in their content of impurities as the major impurities, are (Quartz, feldspar, Mica) (Quartz) (Mica) and (Mica and Quartz) in clinoptonite; analcime; philipsite and chabazaite minerals, respectively. Also, the respective surface areas of such minerals are 407.9, 313.0, 961.7 and 1100 m2 g–1. it is also obvious that, analcime mineral has the lowest CEC and surface area among the used zeolite minerals followed by clinoptonite mineral. This may reflected on the behavior of Pb2+ sorption which will be discussed latter.
Lead adsorption: The kinetics of Pb(II) adsorption were shown by Strawn et al. (1998) at the aluminum oxide-water interface at pH 6.5 to be initially fast, resulting in 76% of the total sorption occurring within 15 min, followed by a slow continuous sorption reaction likely resulting from diffusion through micro pores. Also, Shen et al. (1999) indicated that, all the Pb sorption in clay minerals (as smectite groups) took place within 0.1 h, after which the sorption kinetics exhibited a plateau, a time period of four days was considered adequate to reach equilibrium conditions in pH-edge and isotherm studies.
Pb2+ sorption by the studied clay minerals generally, conformed to the Langmuir sorption isotherm but over limited concentration ranges (Table 2 and Fig. 1). However, at high concentrations of the applied Pb2+, data deviated from the Langmuir model. Such deviations from the Langmuir model indicated that there were different reactions might occurred at higher Pb2+ concentrations especially for analcime and clinoptilolite. The maximum adsorption capacities (i.e., b values) were, 208.33 and 204.08 mg g–1 for phillipsite and chabazite, with correlation coefficient (R2) equal to 0.997 and 0.995, respectively. Also the corresponding binding energy (i.e., Kd) were 2.400 and 0.875 mg L–1, respectively. While for clinoptilolite and analcime the b values were differ in the low and high concentrations of applied Pb2+. The b1 (in low concentrations) reached 88.5 and 100.0 mg g–1 with correlation coefficient (R2) equal to 0.894 and 0.865 for such minerals, respectively (Table 2 and Fig. 1). The differences in b values may be rendered to the large difference between the studied minerals in their contents of Fe and Al oxides as well as, the differences in their impurities content, C.E.C and/or their surface area, (Table 1). Also, the binding energies (Kd) of Pb2+ were high for the studying minerals. This means that the surfaces of zeolite minerals-especially Philipsite and chabazite-strongly adsorbed Pb2+. The maximum adsorption capacities of such minerals for Pb2+ were in the same sequence. This may be due to the higher initial surface area and C.E.C of chabazite and philipsite in (Table 1). In contrast the binding energy (Kd) of analcime was relatively the lowest one among the studying zeolite minerals. Possibly due to its relatively lower cation exchange capacity and surface area, (39 C mol kg–1 and 313.0 m2gm–1, Table 1).
| Table 2: | The Maximum Adsorption Capacity (MAC) and Binding Energy (BE) for the adsorbed Pb2+ in the studying Zeolite minerals according to Langmuir Equation |
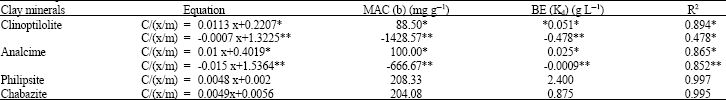 | |
| *Low concentrations and **high concentrations, (R2 was calculated based on 14 points obtained from 7 concentrations of applied Pb in two replicates) | |
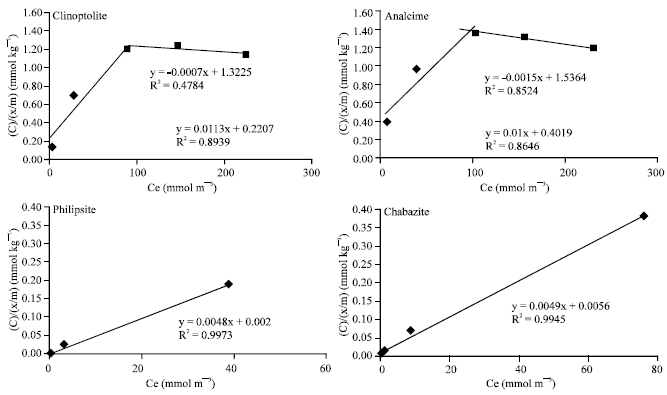 | |
| Fig. 1: | Langmuir adsorption isotherm of Pb2+ in the studying zeolite minerals. (R2 was calculated based on 14 points obtained from 7 concentrations of applied Pb in two replicates) |
| Table 3: | The maximum adsorption capacity (MAC) and binding energy (BE) for the adsorbed Pb2+ in the studying Zeolite minerals according to Frendlich equation |
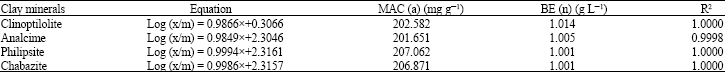 | |
In this respect Gatima et al. (2006), pointed out that the adsorption of lead or lead removal from lead contaminated soils was depended upon the cation exchange capacities of the used red mud, bone meal and pulverized fly ash. As, the CEC could be a major parameter in the adsorption capacity of the soil. The calculating binding energy of Pb2+ with the studied zeolite minerals, show that, in low concentrations of the applied Pb2+, the kd of both analcime and clinoptilolite were so much higher than those at higher concentrations in most cases. This means that, the exchangeable sites adsorbed Pb2+ with relatively higher adsorption energy in low concentrations, more than the available surfaces for the adsorption in higher concentrations. This may be due to the nature of adsorption sites in low concentration. As the adsorbed Pb2+ may be as near as such sites, hence, the adsorption forces could be increased than those under higher concentrations. Generally, the binding energy of the studying zeolite minerals for Pb2+ adsorption could be arranged in the following descending order: philipsite> chabazite>clinoptilolite>analcime. On the other hand, at low concentrations of the applied Pb2+, the b1 values for clinoptilolite reached 88.5 mg g–1. While for analcime it was 100.0 mg g–1. The philipsite and chabazite minerals had not any differences in the obtained b values between higher and lower concentrations of the applied Pb2+. It is obvious that, analcime showed the lowest Pb2+adsorption capacity. This may be due to its relatively lower CEC and surface area as previously mentioned, (Table 1). The used clay minerals could be arranged due to their maximum adsorption capacity for Pb2+ as follows: philipsite> chabazite> clinoptilolite> analcime. The differences between the used clay minerals on their maximum adsorption capacity could be refer to the differences in their impurities content, their CEC and/or their surface area (Table 1). On the other hand, data was applicable to the Freundlich adsorption isotherm over the entire Pb2+ concentration ranges of (0 to 5 μmol mL–1). The binding energy (n) reached, 1.014, 1.005, 1.001 and 1.001 g L–1 for clinoptilolite, analcime, phillipsite and chabazite, respectively. However, the b values (maximum adsorption capacity) reached 202.582, 201.651, 207.062 and 206.871 mg g–1 with correlation coefficient (R2) nearly one for all studying minerals respectively, (Table 3 and Fig. 2).
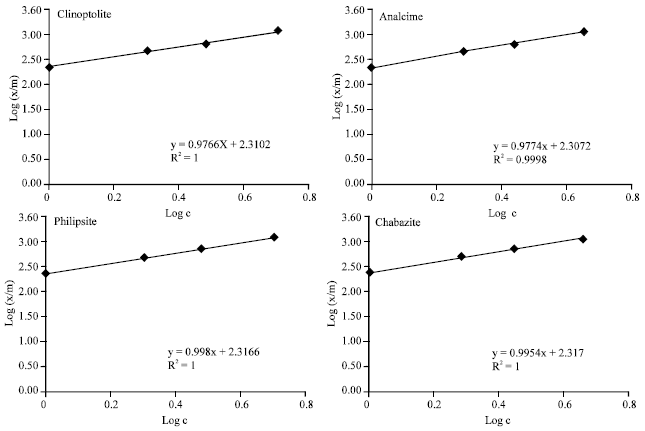 | |
| Fig. 2: | Frendlich adsorption isotherm of Pb2+ in the studying Zeolite minerals. (R2 was calculated based on 14 points obtained from 7 concentrations of applied Pb in two replicates) |
| Table 4: | Adsorption and desorption of Pb2+ on the used zeolite minerals |
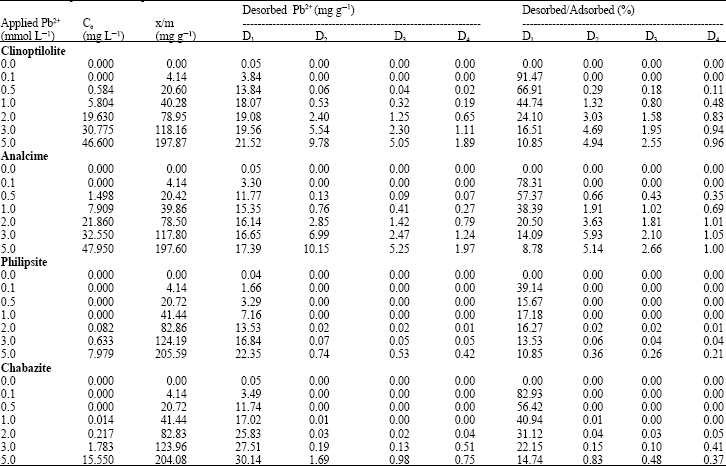 | |
| Where: (Ce) is the equilibrium concentrations in (mg L–1); while (x/m) is the adsorbed amount of Pb2+ in (mg g–1); and (D1, D2, D3 and D4) are the four successive desorptionz | |
It is worth to mention that, the maximum adsorption capacities obtained either with Langmuir or Freundlich models were more or less the same for chabazite and phillipsite. Such results stood in a good agreement with the sorption of lead by clay minerals and goethite, founded by Mario et al. (2002). Generally, the studying Zeolite minerals differ in their ability to adsorb Pb2+ in the following order: philipsite> chabazite> clinoptilolite> analcime. This indicates the profitability of using Zeolite minerals especially, philipsite and chabazite in the field of remediation for the polluted areas as ion exchangers for environmental protection with Pb2+. Also, they could be used for other applications as packing material in subsurface reactive barriers intercepting ground water plumes and for fixed bed reactors designed to remove heavy metals from industrial wastewater. This means that such minerals could play an important role, not only as physical barriers (for the isolation of Pb2+ from metal rich wastes) but also as a chemical barrier, as consequence of the ability of such clay minerals to adsorb lead and to avoid their environmental dispersion of such metal.
Amounts of desorbed Pb2+: Data in Table 4 show the amounts of the extracted Pb2+ in four successive extractions following the Pb2+ adsorption experiment, as well as, percentages of the extracted Pb2+ relative to the amount of adsorbed Pb2+(x/m). Apparently, higher and significantly amounts of extracted Pb2+ were obtained in the first extraction from all studied minerals and then gradually and sharply decreased in the other three extractions. Also, the amounts of desorbed Pb2+ were differed from one mineral to the others. The clinoptilolite and analcime minerals gave the higher amounts of extracted Pb2+. This behavior may be due to the nature of adsorbing sites in such minerals, which may differ than those of chabazite and philipsite. On the other hand, by calculating the desorbed Pb2+ as a percent from the adsorbed Pb2+, one can concluded that, the studied Zeolite minerals were greatly differ than each other. Furthermore, the higher percentages of desorbed lead were obtained in the 1st extraction and then sharply decreased in the following extractions. The differences in such percentages in the 1st extraction were as follows: (10.85-91.47) (8.78-78.31) 10.85-39.14) and (14.74-82.93) for clinoptilolite, analcime, philipsite and chabazite, respectively.
CONCLUSION
The adsorption of lead onto Zeolite minerals has an ion exchange nature. The data are applied well to the Freundlich model, as the correlation coefficient was almost 1.0 for all studied minerals. The amount of ad sorbed Pb2+ increase as the rate of applied Pb2+ increased from 0 to 5 mM L–1 (Fig. 3) for the tested Zeolite minerals.
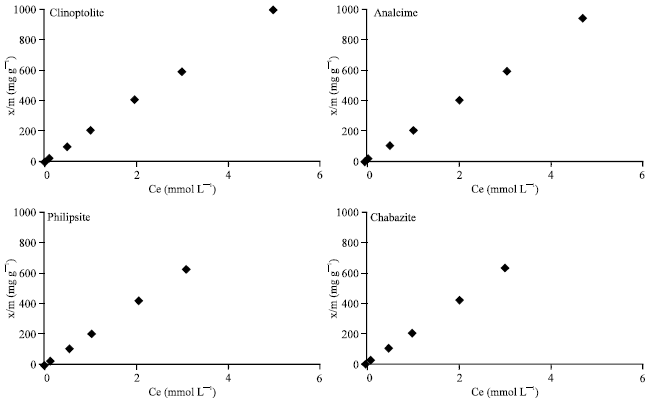 | |
| Fig. 3: | Relationship between the amount of adsorbed Pb2+ by zeolite minerals and the equilibrium concentration of the solution |
The order of Kd values for the used clay minerals were philipsite> chabazite> clinoptilolite> analcime. On the other hand, data agreed very well with langmuir equation, (especially at low concentrations of applied (Pb2+) for clinoptilolite and analcime. Two different stages are observed in the sorption of Pb2+ either by clinoptilolite or analcime, referring to Langmuire model. The process was being with fast adsorption on such Zeolite minerals microcrystal surfaces at low concentrations. Then the Pb2+ sorption was gradually decreased in high concentrations. Such data indicated the efficiency of zeolite materials on Pb2+ sorption. Most of the sorbed Pb2+ was extracted in the 1st extraction following the adsorption experiment. The ability of the used zeolite minerals to retain Pb2+ was high and there were differences between the studied minerals in sorption of Pb2+. This indicates the profitability of using such minerals especially, philipsite and chabazite in the field of remediation for the Pb2+ polluted areas. In conclusion, data indicated that zeolite minerals could be successfully used as a good tool for other applications as packing material in subsurface reactive barriers intercepting ground water plumes and for fixed bed reactors designed to remove Pb2+ from industrial wastewater, however, further researches will be needed to evaluate the efficiency of such zeiolite minerals under the other lead compounds such as lead carbonate, lead sulphate etc.