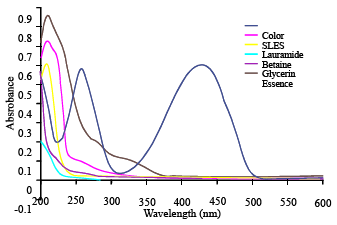
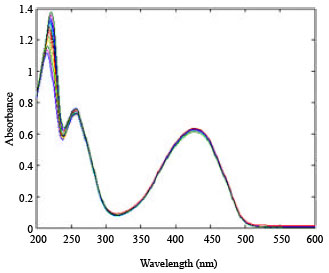
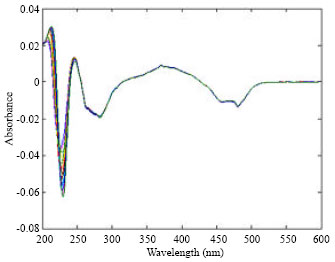
Research Article
Determination of Nonionic Surfactant in Liquid Detergent by UV-vis Spectrophotometry and Multivariate Calibration Method
Department of Chemistry, Azad University-North Tehran Branch, Shariati Street-Zafar Cross Road P.O. Box 19585-936, Tehran, Iran
Arezoo Hassan Nouri
Department of Chemistry, Azad University-North Tehran Branch, Shariati Street-Zafar Cross Road P.O. Box 19585-936, Tehran, Iran
Atena Adnani
Department of Chemistry, Azad University-North Tehran Branch, Shariati Street-Zafar Cross Road P.O. Box 19585-936, Tehran, Iran
Hamidreza Fard Masoumi
Department of Chemistry, Azad University-North Tehran Branch, Shariati Street-Zafar Cross Road P.O. Box 19585-936, Tehran, Iran